Thu, Jul 3, 2025
Volume 17, Issue 1 (Winter & Spring 2020)
ASJ 2020, 17(1): 1-6 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Elyasi L, Ghazvini H. The Protective Effects of Citrus Aurantium Extract on a 6-Hydroxydopamine-Induced Model of Parkinson’s Disease in Male Rats. ASJ 2020; 17 (1) :1-6
URL: http://anatomyjournal.ir/article-1-230-en.html
URL: http://anatomyjournal.ir/article-1-230-en.html
1- Department of Anatomy, Neuroscience Research Center, Faculty of Medicine,Golestan University of Medical Sciences, Golestan, Iran.
2- Department of Neurosience, Immunogenetics Research Center, School of Advanced Technologies in Medicine, Mazandaran University of Medical Sciencess, Mazandaran, Iran.
2- Department of Neurosience, Immunogenetics Research Center, School of Advanced Technologies in Medicine, Mazandaran University of Medical Sciencess, Mazandaran, Iran.
Full-Text [PDF 561 kb]
(1309 Downloads)
| Abstract (HTML) (4645 Views)
Full-Text: (1760 Views)
1. Introduction
Parkinson’s Disease (PD) is a neurodegenerative condition, which damages dopaminergic neurons in the Substantia Nigra pars compacta (SNc). There is inadequate information on the etiology of PD; however, various pathological mechanisms, including lysosomal and mitochondrial dysfunctions, oxidative stress, pathological inclusions, and neuroinflammatory processes have been mentioned [1].
PD can be prevented through regulating intracellular Reactive Oxygen Species (ROS), besides the inhibition of protein aggregation and neuroinflammation [2]. Overall, 6-hydroxydopamine (6-OHDA) is recognized as a common neurotoxin, used to selectively damage dopaminergic neurons [3]. Citrus species are used as medical herbs and food seasoning. These species contain active compounds, such as coumarin, flavonoids, and hesperidins [4]. Among different species, Citrus aurantium (C. aurantium) is a frequently used medicinal plant, endemic to Iran. In traditional medicine, this herb is used to treat and manage neurodegenerative conditions, including seizures, weakness, sleep disorders, pain, and migraine [5].
According to previous studies, C. aurantium has anti-oxidant, anti-cancer, and anti-inflammatory properties. Moreover, flavonoid phytochemical studies have indicated the antioxidant capacity of this plant and its effects on the free-radical reduction of neuronal support metabolism in Alzheimer’s disease [6]. Therefore, we examined the efficacy of C. aurantium extract in protecting against 6-OHDA-mediated oxidative stress [7].
2. Materials and Methods
In this study, 60 male Wistar rats (250-300 g) were obtained from Kerman Animal House. They were kept based on the principles of the Iranian Council of Animal Care. The Ethics Committee of Golestan University approved this study [8]. The animals’ rotational responses to Intraperitoneal (IP) apomorphine injections (2.5 mg/kg in normal saline) were examined. For all the experiments, rats with <30 contralatral turns within 1 h were included. The studied rats were assigned into 6 groups (10 rats per group), as follows: sham (SH); control (C); lesion (L); and CAE-treated lesion (200, 400, and 600 mg/mL CAE+L).
To obtain the extract, the plant was first soaked in methanol. A part of the plant flower was washed and air-dried in a dark place and at room temperature (22-25°C) for 1 week. Briefly, 150 g of the air-dried parts of flower was ground into 50 g of fine powder. Next, 100 g of aerial parts of the C. aurantium powder was mixed with 80% methanol.
Furthermore, to prevent chemical changes under the influence of chemical interactions induced by sunlight radiation on the plant constituents and the evaporation of solvent, the Erlenmeyer flask was covered with foil and its lid with Parafilm. The flask was placed on a shaker at 120 rpm at room temperature for 48 h. The resulting extract was filtered several times with Whatman filter papers to obtain a transparent solution. To remove the organic solvent and increase the concentration of the extract, a distillatory was used in vacuum at 40°C. The required amount of the extract was kept at 4°C; while the remaining extract was driedin an oven at 40°C for 48 h for long-term storage. The latter portion was then stored at 4°C. Figure 1 shows the C. aurantium plant [9].
Ketamine and xylazine (80 and 10 mg/kg, respectively) were injected to anesthetize the rats; then, they were put in a Kopf instrument (Stoelting Instruments, USA). The right side of SNc was determined at the coordinates from the interaural line; the injection site was 3.7, 2.2, and 7.7 mm codal to the interaural line, lateral to the midline, and ventral to the dura, respectively [10]. A Hamilton syringe was used after opening the dura mater. Then, 6-OHDA in 0.2% vitamin C (8 μg/ 2.5 μL) was unilaterally injected to each rat.
The 6-OHDA solution injection was performed gradually at 1 μL/min. The wounds were sutured after the needles remained in place for 5 min before slow removal. The animals received 0.25 mg/kg of apomorphine (Sigma Co, USA) at 2 weeks post-surgery. At 10 min following the injection, the number of rotations was measured for 1 h. The lesion was confirmed if the animal rotated >7 times/min. Additionally, a vehicle (0.9% saline solution with 0.2 mg/mL of l-ascorbate) was administered to the sham group. CAE (200 mg/kg; Tishcon, Osaka, Japan) was orally administered to the L+E group at 1 week before neurotoxin injection, which continued for 2 months [11].
For conducting the apomorphine-induced rotational test, apomorphine hydrochloride (0.5 mg/kg) was used to assess rotational behaviors at one week pre-surgery, as well as two weeks post-surgery. The rotations were determined using a previously introduced method [12]. The rats were accustomed to the experiment during 10 min. Then, complete rotations were measured in a container (height: 35 cm, diameter: 33 cm) at 1 min after the injection in an isolated room for 1 h within 10 min intervals. The positive score minus the negative score indicated the net number of rotations.
For histological analysis, we randomly selected half of the rats from each group. After completing the behavioral experiments, they were deeply anesthetized with 150 mg/kg of ketamine and perfused from the ascending aorta using 0.9% saline (50-100 mL), fixative solution (100-200 mL, 4% paraformaldehyde in phosphate buffer, pH: 7.4), then 100 mL of 0.1 ml phosphate buffer (with 10% sucrose). After removing the animals’ brains following perfusion, brainstem and forebrain blocks were prepared. A freezing microtome (Leica, Germany) was used to cut the sections (50 μm) after preparation in 30% sucrose for 2 to 3 days. Then, they were collected in phosphate buffer (0.1 ml). In addition, 0.1% cresyl violet (Sigma) was used for Nissl staining on every second section.
For mesencephalic sections analysis (2.9-4.2 mm), a method suggested in the literature was applied. Furthermore, stained neurons were manually measured in SNc via light microscopy with a superimposed grid (magnification ×400). At least 2 sections of 4 Paxinos-Watson planes (2.97, 3.2, 3.7, and 4.2) were assessed by scanning each side. The received treatment was not considered in the process of counting [13]. The obtained data are expressed as mean±SEM. Comparisons were made using t-test and one-way Analysis of Variance (ANOVA). P<0.01 was considered as statistically significant.
3. Results
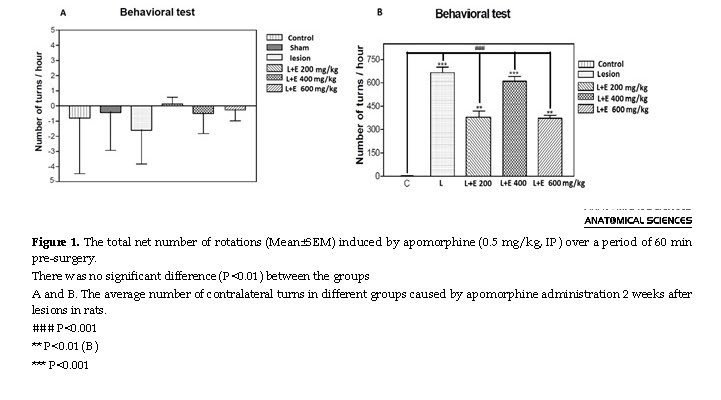
Based on apomorphine-induced rotations analysis, the groups were not significantly different before conducting the surgery. However, the 6-OHDA-lesion group revealed major contralateral rotations in comparison with the sham group at 1 week post-surgery (P<0.001). However, in the first behavioral assessment conducted before surgery, no significant difference was found between the groups (Figure 1A). The C and SH groups were not significantly different in terms of rotation in the second assessment at 2 weeks after surgery. Accordingly, comparisons were made between group C and other groups. In the L and L+E200 mg/kg groups, apomorphine majorly increased contralateral turning (P<0.001). Moreover, it significantly declined the L+E400 mg/kg and L+E600 mg/kg groups versus the L group (Figure 1B).
Parkinson’s Disease (PD) is a neurodegenerative condition, which damages dopaminergic neurons in the Substantia Nigra pars compacta (SNc). There is inadequate information on the etiology of PD; however, various pathological mechanisms, including lysosomal and mitochondrial dysfunctions, oxidative stress, pathological inclusions, and neuroinflammatory processes have been mentioned [1].
PD can be prevented through regulating intracellular Reactive Oxygen Species (ROS), besides the inhibition of protein aggregation and neuroinflammation [2]. Overall, 6-hydroxydopamine (6-OHDA) is recognized as a common neurotoxin, used to selectively damage dopaminergic neurons [3]. Citrus species are used as medical herbs and food seasoning. These species contain active compounds, such as coumarin, flavonoids, and hesperidins [4]. Among different species, Citrus aurantium (C. aurantium) is a frequently used medicinal plant, endemic to Iran. In traditional medicine, this herb is used to treat and manage neurodegenerative conditions, including seizures, weakness, sleep disorders, pain, and migraine [5].
According to previous studies, C. aurantium has anti-oxidant, anti-cancer, and anti-inflammatory properties. Moreover, flavonoid phytochemical studies have indicated the antioxidant capacity of this plant and its effects on the free-radical reduction of neuronal support metabolism in Alzheimer’s disease [6]. Therefore, we examined the efficacy of C. aurantium extract in protecting against 6-OHDA-mediated oxidative stress [7].
2. Materials and Methods
In this study, 60 male Wistar rats (250-300 g) were obtained from Kerman Animal House. They were kept based on the principles of the Iranian Council of Animal Care. The Ethics Committee of Golestan University approved this study [8]. The animals’ rotational responses to Intraperitoneal (IP) apomorphine injections (2.5 mg/kg in normal saline) were examined. For all the experiments, rats with <30 contralatral turns within 1 h were included. The studied rats were assigned into 6 groups (10 rats per group), as follows: sham (SH); control (C); lesion (L); and CAE-treated lesion (200, 400, and 600 mg/mL CAE+L).
To obtain the extract, the plant was first soaked in methanol. A part of the plant flower was washed and air-dried in a dark place and at room temperature (22-25°C) for 1 week. Briefly, 150 g of the air-dried parts of flower was ground into 50 g of fine powder. Next, 100 g of aerial parts of the C. aurantium powder was mixed with 80% methanol.
Furthermore, to prevent chemical changes under the influence of chemical interactions induced by sunlight radiation on the plant constituents and the evaporation of solvent, the Erlenmeyer flask was covered with foil and its lid with Parafilm. The flask was placed on a shaker at 120 rpm at room temperature for 48 h. The resulting extract was filtered several times with Whatman filter papers to obtain a transparent solution. To remove the organic solvent and increase the concentration of the extract, a distillatory was used in vacuum at 40°C. The required amount of the extract was kept at 4°C; while the remaining extract was driedin an oven at 40°C for 48 h for long-term storage. The latter portion was then stored at 4°C. Figure 1 shows the C. aurantium plant [9].
Ketamine and xylazine (80 and 10 mg/kg, respectively) were injected to anesthetize the rats; then, they were put in a Kopf instrument (Stoelting Instruments, USA). The right side of SNc was determined at the coordinates from the interaural line; the injection site was 3.7, 2.2, and 7.7 mm codal to the interaural line, lateral to the midline, and ventral to the dura, respectively [10]. A Hamilton syringe was used after opening the dura mater. Then, 6-OHDA in 0.2% vitamin C (8 μg/ 2.5 μL) was unilaterally injected to each rat.
The 6-OHDA solution injection was performed gradually at 1 μL/min. The wounds were sutured after the needles remained in place for 5 min before slow removal. The animals received 0.25 mg/kg of apomorphine (Sigma Co, USA) at 2 weeks post-surgery. At 10 min following the injection, the number of rotations was measured for 1 h. The lesion was confirmed if the animal rotated >7 times/min. Additionally, a vehicle (0.9% saline solution with 0.2 mg/mL of l-ascorbate) was administered to the sham group. CAE (200 mg/kg; Tishcon, Osaka, Japan) was orally administered to the L+E group at 1 week before neurotoxin injection, which continued for 2 months [11].
For conducting the apomorphine-induced rotational test, apomorphine hydrochloride (0.5 mg/kg) was used to assess rotational behaviors at one week pre-surgery, as well as two weeks post-surgery. The rotations were determined using a previously introduced method [12]. The rats were accustomed to the experiment during 10 min. Then, complete rotations were measured in a container (height: 35 cm, diameter: 33 cm) at 1 min after the injection in an isolated room for 1 h within 10 min intervals. The positive score minus the negative score indicated the net number of rotations.
For histological analysis, we randomly selected half of the rats from each group. After completing the behavioral experiments, they were deeply anesthetized with 150 mg/kg of ketamine and perfused from the ascending aorta using 0.9% saline (50-100 mL), fixative solution (100-200 mL, 4% paraformaldehyde in phosphate buffer, pH: 7.4), then 100 mL of 0.1 ml phosphate buffer (with 10% sucrose). After removing the animals’ brains following perfusion, brainstem and forebrain blocks were prepared. A freezing microtome (Leica, Germany) was used to cut the sections (50 μm) after preparation in 30% sucrose for 2 to 3 days. Then, they were collected in phosphate buffer (0.1 ml). In addition, 0.1% cresyl violet (Sigma) was used for Nissl staining on every second section.
For mesencephalic sections analysis (2.9-4.2 mm), a method suggested in the literature was applied. Furthermore, stained neurons were manually measured in SNc via light microscopy with a superimposed grid (magnification ×400). At least 2 sections of 4 Paxinos-Watson planes (2.97, 3.2, 3.7, and 4.2) were assessed by scanning each side. The received treatment was not considered in the process of counting [13]. The obtained data are expressed as mean±SEM. Comparisons were made using t-test and one-way Analysis of Variance (ANOVA). P<0.01 was considered as statistically significant.
3. Results
Based on apomorphine-induced rotations analysis, the groups were not significantly different before conducting the surgery. However, the 6-OHDA-lesion group revealed major contralateral rotations in comparison with the sham group at 1 week post-surgery (P<0.001). However, in the first behavioral assessment conducted before surgery, no significant difference was found between the groups (Figure 1A). The C and SH groups were not significantly different in terms of rotation in the second assessment at 2 weeks after surgery. Accordingly, comparisons were made between group C and other groups. In the L and L+E200 mg/kg groups, apomorphine majorly increased contralateral turning (P<0.001). Moreover, it significantly declined the L+E400 mg/kg and L+E600 mg/kg groups versus the L group (Figure 1B).
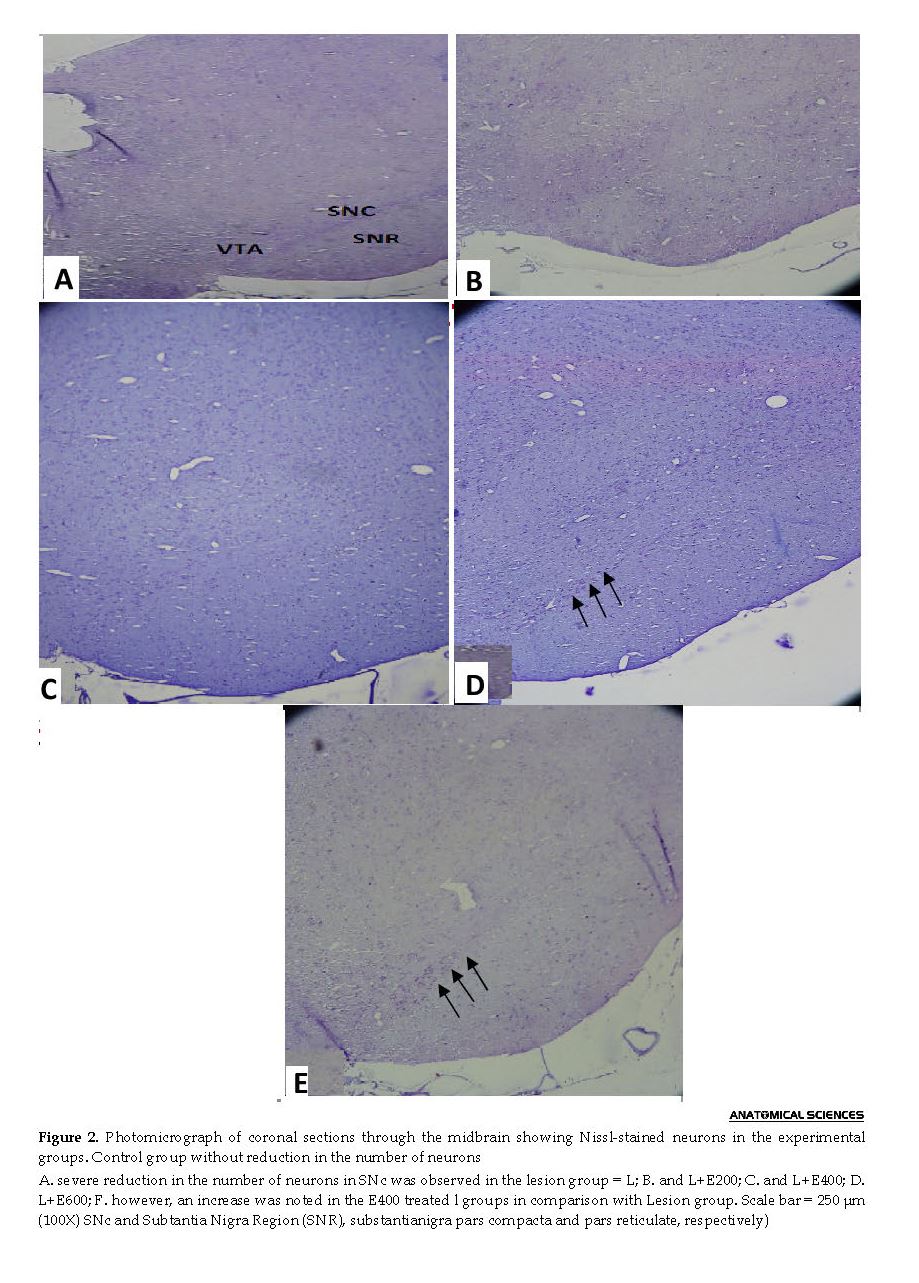
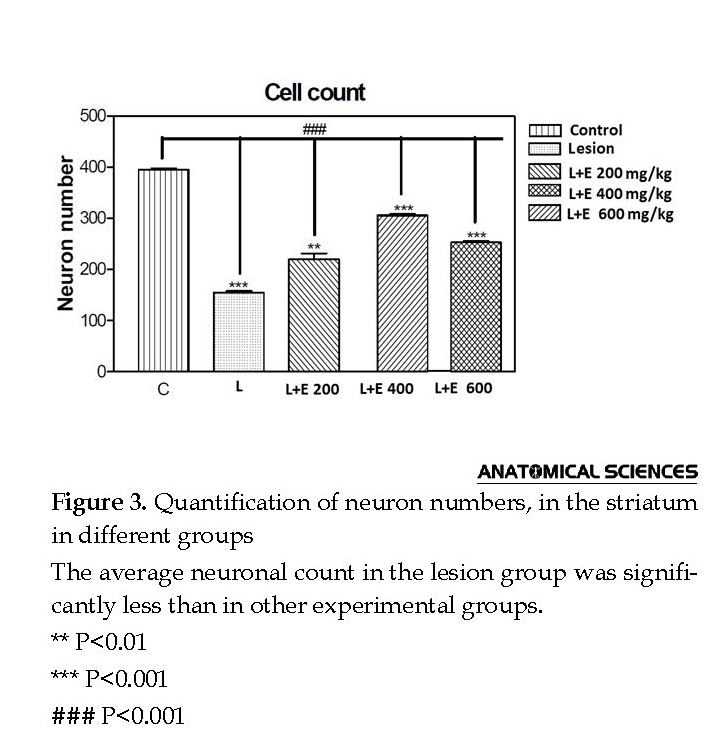
According to the histochemical analyses (Figure 2) and counting (Figure 3), the sham and control groups were not significantly different, considering the stained neuron count on the left side of SNc. Additionally, a significant decline was reported in the 6-OHDA group (lesion = L, P<0.01); while a less prominent decline was observed in the L+E200, L+E400, and L+E600 groups (P<0.05).
4. Discussion
There are no effective treatments for PD as a neurodegenerative disorder. Unilateral 6-OHDA microinjection in the neostriatum of rats produces a reliable and reproducible model of PD for exploring the molecular mechanisms of dopaminergic neuron degeneration and evaluating the efficacy of promising therapeutic candidates [14]. Unilateral disruption (due to intrastriatal 6-OHDA injection) of the nigrostriatal dopaminergic system reduces dopamine inside the striatum and upregulates postsynaptic dopaminergic receptors. According to the present study, these changes in 6-OHDA-lesion rats cause significant motor asymmetry due to contralateral rotations [15]. The lower prevalence of rotational behaviors in the current study might be attributed to the CAE potential in preserving dopaminergic neurons of SNc and keeping striatal dopamine at a suitable level (unrelated to robust motor asymmetry).
4. Discussion
There are no effective treatments for PD as a neurodegenerative disorder. Unilateral 6-OHDA microinjection in the neostriatum of rats produces a reliable and reproducible model of PD for exploring the molecular mechanisms of dopaminergic neuron degeneration and evaluating the efficacy of promising therapeutic candidates [14]. Unilateral disruption (due to intrastriatal 6-OHDA injection) of the nigrostriatal dopaminergic system reduces dopamine inside the striatum and upregulates postsynaptic dopaminergic receptors. According to the present study, these changes in 6-OHDA-lesion rats cause significant motor asymmetry due to contralateral rotations [15]. The lower prevalence of rotational behaviors in the current study might be attributed to the CAE potential in preserving dopaminergic neurons of SNc and keeping striatal dopamine at a suitable level (unrelated to robust motor asymmetry).
C. aurantium seems to produce neuroprotective effects in the primary neurons of mice at concentrations where antioxidant effects are less likely to dominate [16]. Despite its lower estrogenic and antioxidant activity, hesperetin, a vital component of C. aurantium, exerts neuroprotective effects through different pathways (e.g. stimulating tyrosine kinases and estrogenic receptors) [17].
The narrow beam test on 6-OHDA-lesion rats suggested that the time on the beam and latency increased, in comparison with the sham group. Therefore, the depletion of dopamine in the striatum increased the delay in initiating the task and reduced the speed of crossing; i.e. indicative of akinesia or bradykinesia [18]. The frequency of rotations attributed to apomorphine injection reduced in the group. The extract demonstrated protective capacity, which helps keep the dopamine level without causing rotation [19]. CAE, through its antioxidant properties, leads to the functional return of nigroestriatal system [20].
The flavonoids in the extract, including hesperetin, due to free radicals’ collection feature (with anion hydroxyl as a secondary propagulant) activate signaling molecules (e.g. C-protease, and other nuclear factors). By inhibiting ROS production, hesperetin reduces signaling molecules’ activities [21]. The present findings confirmed the therapeutic use of C. aurantium in vivo. This compound was effective in the reversal of behavioral disorders in a PD model. The current findings may provide a new clinical insight into progressive neurodegenerative diseases, like PD.
Ethical Considerations
Compliance with ethical guidelines
The study was performed in accordance with the guidelines of National Institute of Health for the care and use of laboratory animals. The animals were kept based on the principles of the Iranian Council of Animal Care. The Ethics Committee of Golestan University approved this study.
Funding
This study received financial support from the Neuroscience Research Center of Golestan University of Medical Sciences, Iran (grant No: 9411279297).
Authors contributions
All authors have read and approved the manuscript and fulfilled the criteria for authorship according to the ICMJE recommendations for authorship.
Conflict of interest
The authors declared no conflicts of interest.
Acknowledgments
We express our gratitude to our colleagues, who contributed to this research.
References
Eftekhar-Vaghefi S, Esmaeili-Mahani S, Elyasi L, Abbasnejad M. Involvement of mu opioid receptor signaling in the protective effect of opioid against 6-hydroxydopamine-induced SH-SY5Y human neuroblastoma cells apoptosis. Basic and Clinical Neuroscience. 2015; 6(3):171-8. [PMID] [PMCID]
Suryawanshi JA. An overview of citrus aurantium used in treatment of various diseases. African Journal of Plant Science. 2011; 5(7):390-5.
Elyasi L, Eftekhar-Vaghefi SH, Esmaeili-Mahani S. Morphine protects SH-SY5Y human neuroblastoma cells against 6-hydroxydopamine-induced cell damage: Involvement of anti-oxidant, calcium blocking, and anti-apoptotic properties. Rejuvenation Research. 2014; 17(3):255-63. [DOI:10.1089/rej.2013.1473] [PMID]
CaiY, Luo Q, Sun M, Corke H. Antioxidant activity and phenolic compounds of 112 traditional Chinese medicinal plants associated with anticancer. Life Sciences. 2004; 74(17):2157-84. [DOI:10.1016/j.lfs.2003.09.047] [PMID]
Ghasemi K, Ghasemi Y, Ebrahimzadeh MA. Antioxidant activity, phenol and flavonoid contents of 13 citrus species peels and tissues. Pakistan Journal of Pharmaceutical Sciences. 2009; 22(3):277-81. [PMID]
Repetto M, Llesuy S. Antioxidant properties of natural compounds used in popular medicine for gastric ulcers. Brazilian Journal of Medical and Biological Research. 2002; 35(5):523-34. [DOI:10.1590/S0100-879X2002000500003] [PMID]
Kulich SM, Chu CT. Sustained extracellular signal‐regulated kinase activation by 6‐hydroxydopamine: Implications for parkinson’s disease. Journal of Neurochemistry. 2001; 77(4):1058-66. [DOI:10.1046/j.1471-4159.2001.00304.x] [PMID] [PMCID]
Nezhadi A, Ghazi F, Rassoli H, Bakhtiari M, Ataiy Z, Soleimani S, et al. BMSC and CoQ10 improve behavioural recovery and histological outcome in rat model of Parkinson’s disease. Pathophysiology. 2011; 18(4):317-24. [DOI:10.1016/j.pathophys.2011.05.004] [PMID]
Aquino R, Cáceres A, Morelli S, Rastrelli L. An extract of tagetes l ucida and its phenolic constituents as antioxidants. Journal of Natural Product. 2002; 65(12):1773-6. [DOI:10.1021/np020018i] [PMID]
Baluchnejadmojarad T, Roghani M, Nadoushan MRJ, Bagheri M. Neuroprotective effect of genistein in 6‐hydroxydopamine hemi‐parkinsonian rat model. Phytotherapy Research. 2009; 23(1):132-5. [DOI:10.1002/ptr.2564] [PMID]
Roghani M, Niknam A, Jalali-Nadoushan MR, Kiasalari Z, Khalili M, Baluchnejadmojarad T. Oral pelargonidin exerts dose-dependent neuroprotection in 6-hydroxydopamine rat model of hemi-parkinsonism. Brain Research Bulletin. 2010; 82(5):279-83. [DOI:10.1016/j.brainresbull.2010.06.004] [PMID]
Baluchnejadmojarad T, Roghani M, Mafakheri M. Neuroprotective effect of silymarin in 6-hydroxydopamine hemi-parkinsonian rat: Involvement of estrogen receptors and oxidative stress. Neuroscience Letters. 2010; 480(3):206-10. [DOI:10.1016/j.neulet.2010.06.038] [PMID]
Seifhosseini S, Jahanshahi M, Moghimi A, Aazami NS. The effect of scopolamine on avoidance memory and hippocampal neurons in male wistar rats. Basic and Clinical Neuroscience. 2011; 3(1):9-15.
Dauer W, Przedborski S. Parkinson’s disease: Mechanisms and models. Neuron. 2003; 39(6):889-909. [DOI:10.1016/S0896-6273(03)00568-3]
Lundblad M, Usiello A, Carta M, Håkansson K, Fisone G, Cenci MA. Pharmacological validation of a mouse model of l-DOPA-induced dyskinesia. Experimental Neurology. 2005; 194(1):66-75. [DOI:10.1016/j.expneurol.2005.02.002] [PMID]
Ben‐Hur T, Idelson M, Khaner H, Pera M, Reinhartz E, Itzik A, et al. Transplantation of human embryonic stem cell-derived neural progenitors improves behavioral deficit in parkinsonian rats. Stem Cells. 2004; 22(7):1246-55. [DOI:10.1634/stemcells.2004-0094] [PMID]
Del Rio D, Rodriguez-Mateos A, Spencer JP, Tognolini M, Borges G, Crozier A. Dietary (poly) phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxidants & Redox Signaling. 2013; 18(14):1818-92. [DOI:10.1089/ars.2012.4581] [PMID] [PMCID]
Kiasalari Z, Khalili M, Baluchnejadmojarad T, Roghani M. Protective effect of oral hesperetin against unilateral striatal 6-hydroxydopamine damage in the rat. Neurochemical research. 2016; 41(5):1065-72. [DOI:10.1007/s11064-015-1796-6] [PMID]
Ahmad AS, Ansari MA, Ahmad M, Saleem S, Yousuf S, Hoda MN, et al. Neuroprotection by crocetin in a hemi-parkinsonian rat model. Pharmacology Biochemistry & Behavior. 2005; 81(4):805-13. [DOI:10.1016/j.pbb.2005.06.007] [PMID]
Esposito E, Rotilio D, Di Matteo V, Di Giulio C, Cacchio M, Algeri S. A review of specific dietary antioxidants and the effects on biochemical mechanisms related to neurodegenerative processes. Neurobiology of Aging. 2002; 23(5):719-35. [DOI:10.1016/S0197-4580(02)00078-7]
Ferreres F, García‐Viguera C, Tomás‐Lorente F, Tomás‐Barberán FA. Hesperetin: A marker of the floral origin of citrus honey. Journal of the Science of Food and Agriculture. 1993; 61(1):121-3. [DOI:10.1002/jsfa.2740610119]
The flavonoids in the extract, including hesperetin, due to free radicals’ collection feature (with anion hydroxyl as a secondary propagulant) activate signaling molecules (e.g. C-protease, and other nuclear factors). By inhibiting ROS production, hesperetin reduces signaling molecules’ activities [21]. The present findings confirmed the therapeutic use of C. aurantium in vivo. This compound was effective in the reversal of behavioral disorders in a PD model. The current findings may provide a new clinical insight into progressive neurodegenerative diseases, like PD.
Ethical Considerations
Compliance with ethical guidelines
The study was performed in accordance with the guidelines of National Institute of Health for the care and use of laboratory animals. The animals were kept based on the principles of the Iranian Council of Animal Care. The Ethics Committee of Golestan University approved this study.
Funding
This study received financial support from the Neuroscience Research Center of Golestan University of Medical Sciences, Iran (grant No: 9411279297).
Authors contributions
All authors have read and approved the manuscript and fulfilled the criteria for authorship according to the ICMJE recommendations for authorship.
Conflict of interest
The authors declared no conflicts of interest.
Acknowledgments
We express our gratitude to our colleagues, who contributed to this research.
References
Eftekhar-Vaghefi S, Esmaeili-Mahani S, Elyasi L, Abbasnejad M. Involvement of mu opioid receptor signaling in the protective effect of opioid against 6-hydroxydopamine-induced SH-SY5Y human neuroblastoma cells apoptosis. Basic and Clinical Neuroscience. 2015; 6(3):171-8. [PMID] [PMCID]
Suryawanshi JA. An overview of citrus aurantium used in treatment of various diseases. African Journal of Plant Science. 2011; 5(7):390-5.
Elyasi L, Eftekhar-Vaghefi SH, Esmaeili-Mahani S. Morphine protects SH-SY5Y human neuroblastoma cells against 6-hydroxydopamine-induced cell damage: Involvement of anti-oxidant, calcium blocking, and anti-apoptotic properties. Rejuvenation Research. 2014; 17(3):255-63. [DOI:10.1089/rej.2013.1473] [PMID]
CaiY, Luo Q, Sun M, Corke H. Antioxidant activity and phenolic compounds of 112 traditional Chinese medicinal plants associated with anticancer. Life Sciences. 2004; 74(17):2157-84. [DOI:10.1016/j.lfs.2003.09.047] [PMID]
Ghasemi K, Ghasemi Y, Ebrahimzadeh MA. Antioxidant activity, phenol and flavonoid contents of 13 citrus species peels and tissues. Pakistan Journal of Pharmaceutical Sciences. 2009; 22(3):277-81. [PMID]
Repetto M, Llesuy S. Antioxidant properties of natural compounds used in popular medicine for gastric ulcers. Brazilian Journal of Medical and Biological Research. 2002; 35(5):523-34. [DOI:10.1590/S0100-879X2002000500003] [PMID]
Kulich SM, Chu CT. Sustained extracellular signal‐regulated kinase activation by 6‐hydroxydopamine: Implications for parkinson’s disease. Journal of Neurochemistry. 2001; 77(4):1058-66. [DOI:10.1046/j.1471-4159.2001.00304.x] [PMID] [PMCID]
Nezhadi A, Ghazi F, Rassoli H, Bakhtiari M, Ataiy Z, Soleimani S, et al. BMSC and CoQ10 improve behavioural recovery and histological outcome in rat model of Parkinson’s disease. Pathophysiology. 2011; 18(4):317-24. [DOI:10.1016/j.pathophys.2011.05.004] [PMID]
Aquino R, Cáceres A, Morelli S, Rastrelli L. An extract of tagetes l ucida and its phenolic constituents as antioxidants. Journal of Natural Product. 2002; 65(12):1773-6. [DOI:10.1021/np020018i] [PMID]
Baluchnejadmojarad T, Roghani M, Nadoushan MRJ, Bagheri M. Neuroprotective effect of genistein in 6‐hydroxydopamine hemi‐parkinsonian rat model. Phytotherapy Research. 2009; 23(1):132-5. [DOI:10.1002/ptr.2564] [PMID]
Roghani M, Niknam A, Jalali-Nadoushan MR, Kiasalari Z, Khalili M, Baluchnejadmojarad T. Oral pelargonidin exerts dose-dependent neuroprotection in 6-hydroxydopamine rat model of hemi-parkinsonism. Brain Research Bulletin. 2010; 82(5):279-83. [DOI:10.1016/j.brainresbull.2010.06.004] [PMID]
Baluchnejadmojarad T, Roghani M, Mafakheri M. Neuroprotective effect of silymarin in 6-hydroxydopamine hemi-parkinsonian rat: Involvement of estrogen receptors and oxidative stress. Neuroscience Letters. 2010; 480(3):206-10. [DOI:10.1016/j.neulet.2010.06.038] [PMID]
Seifhosseini S, Jahanshahi M, Moghimi A, Aazami NS. The effect of scopolamine on avoidance memory and hippocampal neurons in male wistar rats. Basic and Clinical Neuroscience. 2011; 3(1):9-15.
Dauer W, Przedborski S. Parkinson’s disease: Mechanisms and models. Neuron. 2003; 39(6):889-909. [DOI:10.1016/S0896-6273(03)00568-3]
Lundblad M, Usiello A, Carta M, Håkansson K, Fisone G, Cenci MA. Pharmacological validation of a mouse model of l-DOPA-induced dyskinesia. Experimental Neurology. 2005; 194(1):66-75. [DOI:10.1016/j.expneurol.2005.02.002] [PMID]
Ben‐Hur T, Idelson M, Khaner H, Pera M, Reinhartz E, Itzik A, et al. Transplantation of human embryonic stem cell-derived neural progenitors improves behavioral deficit in parkinsonian rats. Stem Cells. 2004; 22(7):1246-55. [DOI:10.1634/stemcells.2004-0094] [PMID]
Del Rio D, Rodriguez-Mateos A, Spencer JP, Tognolini M, Borges G, Crozier A. Dietary (poly) phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxidants & Redox Signaling. 2013; 18(14):1818-92. [DOI:10.1089/ars.2012.4581] [PMID] [PMCID]
Kiasalari Z, Khalili M, Baluchnejadmojarad T, Roghani M. Protective effect of oral hesperetin against unilateral striatal 6-hydroxydopamine damage in the rat. Neurochemical research. 2016; 41(5):1065-72. [DOI:10.1007/s11064-015-1796-6] [PMID]
Ahmad AS, Ansari MA, Ahmad M, Saleem S, Yousuf S, Hoda MN, et al. Neuroprotection by crocetin in a hemi-parkinsonian rat model. Pharmacology Biochemistry & Behavior. 2005; 81(4):805-13. [DOI:10.1016/j.pbb.2005.06.007] [PMID]
Esposito E, Rotilio D, Di Matteo V, Di Giulio C, Cacchio M, Algeri S. A review of specific dietary antioxidants and the effects on biochemical mechanisms related to neurodegenerative processes. Neurobiology of Aging. 2002; 23(5):719-35. [DOI:10.1016/S0197-4580(02)00078-7]
Ferreres F, García‐Viguera C, Tomás‐Lorente F, Tomás‐Barberán FA. Hesperetin: A marker of the floral origin of citrus honey. Journal of the Science of Food and Agriculture. 1993; 61(1):121-3. [DOI:10.1002/jsfa.2740610119]
Type of Study: Original |
Subject:
Neuroanatomy
Received: 2019/02/10 | Accepted: 2019/09/10 | Published: 2020/01/1
Received: 2019/02/10 | Accepted: 2019/09/10 | Published: 2020/01/1
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |