Tue, Jul 8, 2025
Volume 18, Issue 1 (Winter & Spring 2021)
ASJ 2021, 18(1): 15-22 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Bahadoran H, Asadi M H, Soltan Hosseini R, Jalali Kondori B. The Effects of Bioadhesive Wound Healer (AMONIA) on Skin Wound Healing. ASJ 2021; 18 (1) :15-22
URL: http://anatomyjournal.ir/article-1-277-en.html
URL: http://anatomyjournal.ir/article-1-277-en.html
1- Department of Anatomical Sciences, School of Medical Sciences, Baqiyatallah University of Medical Sciences, Tehran, Iran.
2- Department of Veterinary Medicine, School of Veterinary Medicine, Shahrekord Branch, Islamic Azad University, Shahrekord, Iran.
3- Baqiyatallah Research Center for Gastroenterology and Liver Diseases, Baqiyatallah University of Medical Sciences, Tehran, Iran.
2- Department of Veterinary Medicine, School of Veterinary Medicine, Shahrekord Branch, Islamic Azad University, Shahrekord, Iran.
3- Baqiyatallah Research Center for Gastroenterology and Liver Diseases, Baqiyatallah University of Medical Sciences, Tehran, Iran.
Full-Text [PDF 1381 kb]
(860 Downloads)
| Abstract (HTML) (2370 Views)
Full-Text: (1200 Views)
1. Introduction
Askin wound is a type of dermal injury which occurs relatively quickly in which skin is torn, cut, or punctured (an open wound) or where blunt force trauma causes a contusion (a closed wound). In pathology, it specifically refers to an acute injury that damages the epidermis of the skin. The body undertakes a series of actions, collectively known as the wound healing process. Healing can be accelerated by applying exogenous substances, like AMONIA [1].
Wound healer bioadhesives, the AMONIA, are considered crucial compounds in various medical fields, like dermatology. These materials prevent bleeding, increase the rate of tissue repair, tissue flexibility, tissue adhesion, cell-cell interactions, surface coherence, cell-cell cohesion, tissue resistance to detergents, and the mechanism of drug release. Due to their specific characteristics, such as high-speed application and fast healing rate with less pain, these tissue adhesives can be considered an appropriate alternative agent for surgical sutures in animals. Moreover, due to their healing properties for injuries, these bioadhesive compounds could increase the healing rate, reduce recovery time, infection, hospitalization costs, tissue scars, and antibiotics consumption in animals. For soft tissue applications, different formulations of bioadhesives have been developed. Moreover, their clinical use remains limited by inadequate well-known mechanisms. Thus, the modern tissue adhesive agents with proper adhesion, adequate tensile strength, as well as high speed and quality of wound healing have been considered in numerous investigations [2].
Bioadhesives and bioremediators present various unique properties, as follows: functional independence; no needs for surface preparation for drug activity; vast application at higher or lower temperatures of the environment; the lack of moisture absorption; high adhesion strength to wet surfaces [3-6]; the ability of binding to various kinds of materials or tissues, high speed and facility of application [7]; hemorrhage prevention; no blood circulation disturbances; inflammatory reactions inhibition; no tissues and organisms movement disturbances; degradability, and the ability to be sterilized. Wound healer bioadhesive (AMONIA, Behzist Irik Novin Daroo Company, Isfahan, Iran) was experimentally used in several livestocks. AMONIA is a miracle among the ranchers, i.e., topically applied as a powder to the excisional wounds without tissue. The composition of AMONIA powder contains amino sugars, sponge extract, magnesium, and galactosamine, as well as various mineral compounds. These materials are responsible for the probable therapeutic features of AMONIA.
No effective wound healing drugs are established; thus, this study aimed to evaluate the bioadhesive wound healing effects of AMONIA on skin wounds created on the white male rabbits, compared to 1% phenytoin ointment [8]. The study samples were analyzed using microbiological and macroscopic assessments.
2. Materials and Methods
This experimental study was performed on 10 adult male rabbits (3860-4400 g, 12-month-olds). All explored animals were kept in standard living conditions, including; 20-23oC temperature, 12:12 light-dark photo-cycle, access to water and food ad libitum. Three wounds (1×1 cm) were created on the back of each study animal (N=30 wounds). Epidermis and dermis were totally excised (4mm depth). They were randomly divided into 3 wound groups, as follows: the negative control group received surgical excision without treatment, the positive control group was treated by phenytoin 1% ointment (Darupakhsh company), and the AMONIA group was treated with AMONIA gel. Finally, the wounds were bandaged; accordingly, they were evaluated daily in all study groups regarding infection, macroscopic degenerative changes, and morphometric features. All treatment procedures were applied daily (at 10 AM) for 22 consecutive days [8, 9].
Intraperitoneal injections of ketamine (70 mg/kg) and xylazine (8 mg/kg) were employed to induce anesthesia. Round skin wounds (1×1 cm) were generated following shaving the animals’ back hair and disinfection with alcohol 70% using a scalpel. Wound depth was considered as full dermis and epidermis thickness (4 mm). The day of creating the surgical wound was considered day 1, followed by treatment processes (AMONIA & phenytoin 1%).
Wound healer adhesives were prepared as a powder (Behzist Airic Novin Daru, Isfahan, Scientific Research Town, Iran, registration No: 1723). This powder had milky color and specific odor and w:as char:acterized by physical and chemical features. The AMONIA powder was used according to the company’s instructions, as follows: 1 gr of AMONIA (1 gr/1×1cm wound) was dissolved in distilled water (2 mL) to form gel treatment. The gel was topically administrated in skin wounds.
To assess the wound healing, wound surface (using a caliber), healing rate (using the formula), and time required for total wound closure (from the first day of wound creation to total skin healing) were recorded. Wound surface (mm2) was measured from the first day of the experiment and every even day from 2-22 based on the Cumming protocol [9]. Wound healing was calculated by the following formula: The percentage of wound area = wound length on the day of measurement/wound length on the first day × 100, and percentage of wound healing = 100 – the percentage of the wound area. All wound creation procedures, measuring wound surface and the studied animals’ weight were applied at a specific time (10 AM) by a fixed individual. Additionally, the wounds were examined by a surgeon on days 3, 7, 14, 18, and 22 regarding the infection, scar, and other factors. Following the preparation of digital images, the Image-J software (the USA, 2015) was used to macroscopically analyze the size and area of the wound [8].
Soyabean Casein Digest Agar and Sabouraud dextrose agar culture media were used to study the aerobic bacteria and yeasts, respectively. Microorganism assessment was defined by the plate-count method. By the Seed Lot technique in sodium chloride-peptone solution buffer culture medium (pH 7.0), the dilution with 1/10 concentrations was prepared and followed by filtration (<45 μm) assay. The filter was transferred to Soyabean Casein Digest Agar solid and Sabouraud dextrose agar solid culture media to analyze aerobic bacteria and yeasts, respectively. These measures were followed by incubation at 25-30°C for 7-14 days. Two series of laboratory analyses were performed for each research sample; the first probable presence of pathogenic bacteria and fungi in bioadhesive healer (authorized in the European Pharmacopoeia for wound healer compounds) was assessed. Next, the second various culture media (including Muller-Hinton Eosin Methylene Blue Blood, and Agar; Germany Merck company) were hired to evaluate the antibacterial effects of AMONIA. On days 3, 6, 11, 14, and 20 of each treatment group, the wound samples of rabbits were randomly collected and cultured. Following 24 and 48 hours of incubation (37°C), the smear was prepared from expanded colonies. Subsequently, Gram staining was performed to evaluate the shape and stainability features. To evaluate the antimicrobial effects of AMONIA, various concentrations (20%, 40%, 60%, 80%, & 100%) of this material were applied on the common bacteria of wound infection, including Staphylococcus aureus, Escherichia coli, and Pseudomonas aeruginosa (respectively with the codes of 1337, 1272, & 1073). The bacteria were selected from the collection of scientific and industrial bacteria and fungi of Iran. The obtained results were reported as growth or non-growth phases, compared to positive and negative controls using the Kirby-Borrower antibiogram method. The disc in the negative control group had no active substance; the disk in the positive control had ciprofloxacin, i.e., used for Staphylococcus aureus and Escherichia coli; the disc with gentamicin was used for Pseudomonas aeruginosa (European pharmacopoeia) assessment.
The obtained data were analyzed using SPSS v. 12. One-way Analysis of Variance (ANOVA) was employed to determine the between-group differences. Student Samples t-test was also implemented to assess the differences between the two groups. The collected results were reported as Mean±SEM at the significance level of P<0.05 was.
3. Results
Significantly (P<0.05), the rate of wound healing was detected to be 15%, 52%, 74%, 97%, and 100%, respectively in the treatment days of 2, 4, 6, 8, and 10 in the AMONIA group, compared to the positive and negative controls. On the fourth day of treatment in the AMONIA group, the wound surface was more clearly different (completely visually recognizable) than the other groups. However, in the positive control group, no complete recovery was found until the treatment day of 22 Besides, the percentage of wound healing was detected as 68% on the day of 22 in the negative control animals (Table 1).
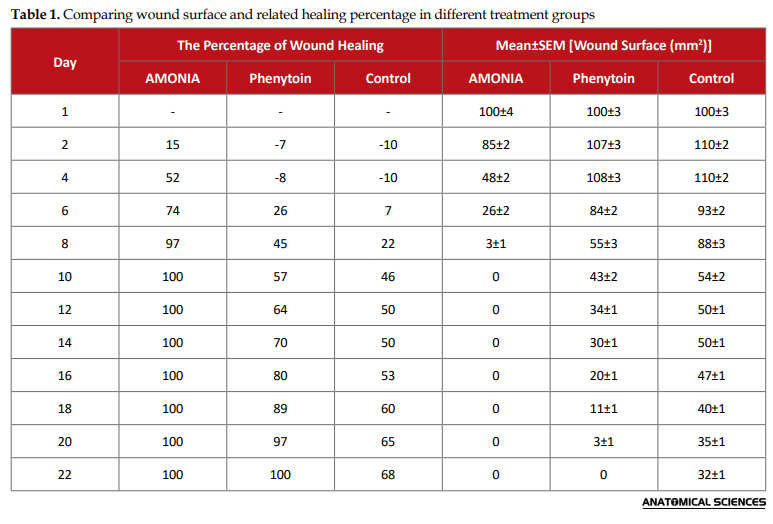
Thus, the total recovery rate of injured skin was detected in the AMONIA group following 10 days of treatment. Furthermore, this value was identified in the Phenytoin group on the day of 22. The Mean±SEM time required for total wound healing in the negative control group treated with phenytoin and the AMONIA group were 22±3, 19.3±1.5, and 9±2 days, respectively. Therefore, there existed significant (P<0.05) differences between the study groups. The primary wound surface was approximately 100 mm2 immediately after dermal incision creation. A significant (P<0.05) decreased level of wound surface was found in the AMONIA group, compared to the control groups. In the AMONIA group, the wound was entirely generated on the 10th day of treatment; however, this value in the control group was equal to 32±1 mm2, representing the therapeutic effects of AMONIA on wound healing.
At the end of the treatment, the animals’ Mean±SEM weight was calculated as 3260±15, 3340±15, and 3380±15 g, respectively; however, no significant (P>0.05) differences were observed between the control and treatment groups. This comparison was also assessed on other treatment days; however, no significant (P>0.05) difference was found in this respect.
This technique indicated that the AMONIA was initially without Escherichia coli, Pseudomonas aeruginosa, and Staphylococcus aureus. Additionally, this substance caused a growth inhibition zone in all concentrations (20%, 40%, 60%, 80%, & 100%), representing the antimicrobial effects of AMONIA. Thus, the Mean±SEM diameter of growth inhibition zone for Staphylococcus, Escherichia coli, and Pseudomonas aeruginosa were computed as 13±2 mm2, 18±3 mm2, and 20±3 mm2, respectively. These findings indicated the inhibitory effects of this substance on common bacterial growth available in the wound site. The microbiological assessment of wound site on days 2, 5, 8, 11, 14, 17, and 20 in the control group revealed several gram-negative bacilli, negative and gram-positive cocci, and yeast. Furthermore, in the positive control group, gram-negative bacilli, gram-negative coccobacillus, and gram-positive cocci were observed.
Various macroscopic assessments were found on the daily observation of skin wounds in the treatment and control groups. As demonstrated in Figure 1, three types of skin wounds were created on the back of the animals, including negative control wound (black arrows), positive control wound (blue arrows), and AMONIA (yellow arrows). On the second day of treatment (Figure 1A), a wide-open wound with bleeding was found in the negative control group (Figure 1A, black arrow); however, the wound in the positive control represented the wound contraction (Figure 1A, blue arrow), leading to coagulation. Moreover, in the last treatment group, the AMONIA gel was applied to the skin which caused wound contraction and blood coagulation (Figure 1A, yellow arrow). On the fourth day of treatment (Figure 1B), although the coagulation was found in the negative and positive control groups (Figure 1B, black & blue arrow), in the AMONIA group (Figure 1B, yellow arrow), no coagulation was detected, leading to decreased inflammation. On the eighth day of treatment (Figure 1C), the negative control group (Figure 1C, black arrow) represented not the healing condition, compared to the AMONIA group (Figure 1C, yellow arrow); the positive control treated with phenytoin presented 15% healing rate; finally, the AMONIA treatment group (Figure 1C, yellow arrow) presented 97% dermal regeneration, compared to the controls.
.PNG)
4. Discussion
Wounds are among the superficial tissue injuries that can be healed quickly without crucial complications. However, this condition can potentially shift to a harmful pathological status that ultimately impairs tissue function. Thus, managing wound progression in the early stages of injury seems necessary to prevent further unpredictable outcomes. Applying AMONIA in acute injuries can represent effective physiological features. In this study, skin wounds were created in laboratory male rabbits and treated with AMONIA. Several factors were studied in treated wounds to evaluate the effects of this drug on wound healing in the primary stages of injury [1].
AMONIA is a natural product used for treating acute skin injuries. This substance is used immediately after acute skin wounds. AMONIA is a powder that could turn into a white gel following combination with sterile distilled water. This gel is topically applied to animals’ wounds to prevent bleeding and accelerate wound healing.
The essential properties of AMONIA belong to the presence of some natural substances, such as glycosaminoglycan, sponge extract, and magnesium. Glycosaminoglycans are long linear polysaccharides, consisting of repeating disaccharide units. Glycosaminoglycans are large molecules that quickly absorb water due to the presence of different carbohydrate classes [10]. By combining glycosaminoglycan powder with distilled water, a hydrophilic gel is immediately formed, which binds temporarily to the surrounding tissue structures. The formed gel has a high viscosity between two sterile media inside the body and a non-sterile medium outside. This agent acts as a barrier that maintains the sterility of the wound and prevents infection. Thus, in the present study, no cases of infection were found in the examined animals treated with AMONIA. Moreover, the prepared hydrogel has a high viscosity, which prevents the passage of microorganisms into the wound site; accordingly, it acts as the next characteristic in maintaining wound sterility. Sponge extract is among the natural factors in ammonia powder. This extract presents potential therapeutic properties against the activity of microorganisms, like bacteria. Sponge extract penetrates the internal environment of the bacterium and prevents proliferation; thus, it inhibits the growth of infection [11]. Magnesium is a trace element effective in multiple biological activities. A deficiency of this substance in the body causes functional defects in the cells of the immune system; therefore, magnesium plays an active role in the activity of the immune system to deal with pathogens. Magnesium plays an essential role in the respiratory bursts of immune cells in hypoxic environments. This trace element increases the rate of phagocytosis in macrophages and induces the exocytosis of immune granules. In this experimental investigation, no infection was observed in the AMONIA group. Such findings could be due to the presence of high-dose magnesium leading to the accelerated activity of immune cells available at the wound site [12].
Following exposure to blood or plasma, it was activated with an adhesive feature after approximately 1-2 minutes. Thus, the bleeding rate was reduced. The evaluated wound manifested no requiring dressing and interference with the study animals’ movements. Besides, the level of scar formation was low in the treatment sample, compared with the controls. This stage can lead to arrest in bleeding and significantly reduces the migration of immune cells to the wound site. As mentioned earlier, this type of hydrogel with antibacterial properties inhibit the presence of infection in two stages; due to its high viscosity of AMONIA hydrogel, this substance prevents the penetration of microorganism into the wound as well as exogenous microorganisms to be proliferated in wounds.
The time required for total wound healing in the AMONIA group was 13 days less than the positive control group (phenytoin group). This study revealed that AMONIA has strong antimicrobial properties, at least on Pseudomonas aeruginosa and Escherichia coli. These bacteria are the main cause of wound infection and among the most resistant gram-negative bacilli to antibiotics and chemical disinfectants. The inhibitory effects of AMONIA on coagulase-positive cocci, like Staphylococcus aureus is also of scientific interest. According to the obtained results, wound healer bioadhesive (AMONIA) accelerated wound healing, especially in the fourth to eighth treatment days. Accordingly, the wound surface and time required for recovery were reduced, and the healing rate was enhanced. Due to the mechanisms involved in skin wound healing, the AMONIA could accelerate wound healing by reducing swelling or inflammation. In other words, by modulating the inflammatory phase [13], the wound healing rate could be accelerated [14]. The prevention of wound infection and keeping the sterility of wound surface can also increase the rate of wound healing. As per the obtained data, antibiotics can control wound infections leading to wound recovery [15]. It was found that the rate of wound healing is not similar in phenytoin and AMONIA groups. Thus, the proposed phenytoin mechanisms involved in wound healing acceleration included the increased concertation of growth factors [16]; macrophages migration [17]; wound elasticity; new vascular formation; collagen synthesis, and fibroblast infiltration [18]. These potential mechanisms should be carefully evaluated.
Overall, for the first time, this study signified that the AMONIA could accelerate skin wound healing in rabbits. Additionally, the AMONIA efficacy in wound healing was found to be more effective than phenytoin 1% ointment. Thus, clinical trials in humans are recommended to find the potential mechanisms mentioned earlier, such as the effects on collagen synthesis; reduced inflammation; increased oxygen delivery; increased angiogenesis, and fibroblast filtration.
In 2007, the application of cyanoacrylate on the skin sutures of rats was accessed. This study indicated that cyanoacrylate, i.e., considered a tissue adhesive agent, is well accepted by the tissue without immunological reaction and inflammation. It was concluded that cyanoacrylate has more advantages than surgical sutures, including complexity and cost-effectiveness [19].
Hossein Nazari et al. evaluated the effects of alcoholic and aqueous extracts of oatmeal on wound healing and skin inflammation. They identified the therapeutic effects of alcoholic and aqueous extracts of oatmeal, which reduced treatment time from 11 to 4 days [20]. Conducting an experimental study between the alcoholic extract of oats and this bioremediation is strictly recommended. Various research studies have been conducted in the field of wound healing; accordingly, various healer substances have been prepared and introduced on an herbal and chemical basis. These studies include the effects of calcium, copper, and zinc ions [21, 22]; physical forces, such as radiation, electrical stimulation [23] ultrasound A waves [24], and chemicals, such as hydrocortisol [25], vitamins [26], ascorbic acid [27], phenytoin [8], saline [28], growth factors [29], and honey [26, 30].
5. Conclusion
According to the present study findings, AMONIA, through various molecular mechanisms, can inhibit tissue bleeding leading to the prevention of inflammation. This phenomenon can lead to a decreased rate of tissue damage. Additionally, this substance includes various natural substances, leading to wound healing acceleration. According to the current research findings, applying AMONIA seems useful for animal care; however, extensive efforts are required for performing human clinical trials concerning AMONIA administration.
Ethical Considerations
Compliance with ethical guidelines
All experimental procedures were conducted under the supervision of the Ethics Committee of Falavarjan University of Medical Sciences (Code: IR.FUMS.REC.1397.065).
Funding
This research did not receive any grant from funding agencies in the public, commercial, or non-profit sectors.
Authors' contributions
Conceptualization and supervision: Mohammad Hossein Asadi; Methodology: Reza Soltan Hosseini; Investigation, writing – original draft, and writing – review & editing: All authors; Data collection: Hosein Bahadoran, and Bahman Jalali Kondori; Data analysis: Bahman Jalali Kondori and Reza Soltan Hosseini.
Conflict of interest
The authors declared no conflicts of interest.
Acknowledgments
We acknowledge the Vice Chancellor for Research (Islamic Azad University, Falavarjan Branch), Dr. Hamid Fooladgar and his colleagues in the research laboratory, as well as the board members of Behzist Airik Novin Daroo (Behzist Airic Novin Daru, Iran) company; Dr. Mostafa Mohammadi, Mr. Haj Agha Mirdamadi, and Mr. Haj Agha Nasher Al-Ahkami for their support. We also acknowledge Ms. Mohadeseh Masaeli, Bahareh Sholeh, Mojgan Afshari, Maryam Ghasemi (the experts of the Islamic Azad University of Falavarjan), and Dr. Mohammad Reza Ebad (a veterinarian and the technical manager of the company) for their technical support.
Askin wound is a type of dermal injury which occurs relatively quickly in which skin is torn, cut, or punctured (an open wound) or where blunt force trauma causes a contusion (a closed wound). In pathology, it specifically refers to an acute injury that damages the epidermis of the skin. The body undertakes a series of actions, collectively known as the wound healing process. Healing can be accelerated by applying exogenous substances, like AMONIA [1].
Wound healer bioadhesives, the AMONIA, are considered crucial compounds in various medical fields, like dermatology. These materials prevent bleeding, increase the rate of tissue repair, tissue flexibility, tissue adhesion, cell-cell interactions, surface coherence, cell-cell cohesion, tissue resistance to detergents, and the mechanism of drug release. Due to their specific characteristics, such as high-speed application and fast healing rate with less pain, these tissue adhesives can be considered an appropriate alternative agent for surgical sutures in animals. Moreover, due to their healing properties for injuries, these bioadhesive compounds could increase the healing rate, reduce recovery time, infection, hospitalization costs, tissue scars, and antibiotics consumption in animals. For soft tissue applications, different formulations of bioadhesives have been developed. Moreover, their clinical use remains limited by inadequate well-known mechanisms. Thus, the modern tissue adhesive agents with proper adhesion, adequate tensile strength, as well as high speed and quality of wound healing have been considered in numerous investigations [2].
Bioadhesives and bioremediators present various unique properties, as follows: functional independence; no needs for surface preparation for drug activity; vast application at higher or lower temperatures of the environment; the lack of moisture absorption; high adhesion strength to wet surfaces [3-6]; the ability of binding to various kinds of materials or tissues, high speed and facility of application [7]; hemorrhage prevention; no blood circulation disturbances; inflammatory reactions inhibition; no tissues and organisms movement disturbances; degradability, and the ability to be sterilized. Wound healer bioadhesive (AMONIA, Behzist Irik Novin Daroo Company, Isfahan, Iran) was experimentally used in several livestocks. AMONIA is a miracle among the ranchers, i.e., topically applied as a powder to the excisional wounds without tissue. The composition of AMONIA powder contains amino sugars, sponge extract, magnesium, and galactosamine, as well as various mineral compounds. These materials are responsible for the probable therapeutic features of AMONIA.
No effective wound healing drugs are established; thus, this study aimed to evaluate the bioadhesive wound healing effects of AMONIA on skin wounds created on the white male rabbits, compared to 1% phenytoin ointment [8]. The study samples were analyzed using microbiological and macroscopic assessments.
2. Materials and Methods
This experimental study was performed on 10 adult male rabbits (3860-4400 g, 12-month-olds). All explored animals were kept in standard living conditions, including; 20-23oC temperature, 12:12 light-dark photo-cycle, access to water and food ad libitum. Three wounds (1×1 cm) were created on the back of each study animal (N=30 wounds). Epidermis and dermis were totally excised (4mm depth). They were randomly divided into 3 wound groups, as follows: the negative control group received surgical excision without treatment, the positive control group was treated by phenytoin 1% ointment (Darupakhsh company), and the AMONIA group was treated with AMONIA gel. Finally, the wounds were bandaged; accordingly, they were evaluated daily in all study groups regarding infection, macroscopic degenerative changes, and morphometric features. All treatment procedures were applied daily (at 10 AM) for 22 consecutive days [8, 9].
Intraperitoneal injections of ketamine (70 mg/kg) and xylazine (8 mg/kg) were employed to induce anesthesia. Round skin wounds (1×1 cm) were generated following shaving the animals’ back hair and disinfection with alcohol 70% using a scalpel. Wound depth was considered as full dermis and epidermis thickness (4 mm). The day of creating the surgical wound was considered day 1, followed by treatment processes (AMONIA & phenytoin 1%).
Wound healer adhesives were prepared as a powder (Behzist Airic Novin Daru, Isfahan, Scientific Research Town, Iran, registration No: 1723). This powder had milky color and specific odor and w:as char:acterized by physical and chemical features. The AMONIA powder was used according to the company’s instructions, as follows: 1 gr of AMONIA (1 gr/1×1cm wound) was dissolved in distilled water (2 mL) to form gel treatment. The gel was topically administrated in skin wounds.
To assess the wound healing, wound surface (using a caliber), healing rate (using the formula), and time required for total wound closure (from the first day of wound creation to total skin healing) were recorded. Wound surface (mm2) was measured from the first day of the experiment and every even day from 2-22 based on the Cumming protocol [9]. Wound healing was calculated by the following formula: The percentage of wound area = wound length on the day of measurement/wound length on the first day × 100, and percentage of wound healing = 100 – the percentage of the wound area. All wound creation procedures, measuring wound surface and the studied animals’ weight were applied at a specific time (10 AM) by a fixed individual. Additionally, the wounds were examined by a surgeon on days 3, 7, 14, 18, and 22 regarding the infection, scar, and other factors. Following the preparation of digital images, the Image-J software (the USA, 2015) was used to macroscopically analyze the size and area of the wound [8].
Soyabean Casein Digest Agar and Sabouraud dextrose agar culture media were used to study the aerobic bacteria and yeasts, respectively. Microorganism assessment was defined by the plate-count method. By the Seed Lot technique in sodium chloride-peptone solution buffer culture medium (pH 7.0), the dilution with 1/10 concentrations was prepared and followed by filtration (<45 μm) assay. The filter was transferred to Soyabean Casein Digest Agar solid and Sabouraud dextrose agar solid culture media to analyze aerobic bacteria and yeasts, respectively. These measures were followed by incubation at 25-30°C for 7-14 days. Two series of laboratory analyses were performed for each research sample; the first probable presence of pathogenic bacteria and fungi in bioadhesive healer (authorized in the European Pharmacopoeia for wound healer compounds) was assessed. Next, the second various culture media (including Muller-Hinton Eosin Methylene Blue Blood, and Agar; Germany Merck company) were hired to evaluate the antibacterial effects of AMONIA. On days 3, 6, 11, 14, and 20 of each treatment group, the wound samples of rabbits were randomly collected and cultured. Following 24 and 48 hours of incubation (37°C), the smear was prepared from expanded colonies. Subsequently, Gram staining was performed to evaluate the shape and stainability features. To evaluate the antimicrobial effects of AMONIA, various concentrations (20%, 40%, 60%, 80%, & 100%) of this material were applied on the common bacteria of wound infection, including Staphylococcus aureus, Escherichia coli, and Pseudomonas aeruginosa (respectively with the codes of 1337, 1272, & 1073). The bacteria were selected from the collection of scientific and industrial bacteria and fungi of Iran. The obtained results were reported as growth or non-growth phases, compared to positive and negative controls using the Kirby-Borrower antibiogram method. The disc in the negative control group had no active substance; the disk in the positive control had ciprofloxacin, i.e., used for Staphylococcus aureus and Escherichia coli; the disc with gentamicin was used for Pseudomonas aeruginosa (European pharmacopoeia) assessment.
The obtained data were analyzed using SPSS v. 12. One-way Analysis of Variance (ANOVA) was employed to determine the between-group differences. Student Samples t-test was also implemented to assess the differences between the two groups. The collected results were reported as Mean±SEM at the significance level of P<0.05 was.
3. Results
Significantly (P<0.05), the rate of wound healing was detected to be 15%, 52%, 74%, 97%, and 100%, respectively in the treatment days of 2, 4, 6, 8, and 10 in the AMONIA group, compared to the positive and negative controls. On the fourth day of treatment in the AMONIA group, the wound surface was more clearly different (completely visually recognizable) than the other groups. However, in the positive control group, no complete recovery was found until the treatment day of 22 Besides, the percentage of wound healing was detected as 68% on the day of 22 in the negative control animals (Table 1).

Thus, the total recovery rate of injured skin was detected in the AMONIA group following 10 days of treatment. Furthermore, this value was identified in the Phenytoin group on the day of 22. The Mean±SEM time required for total wound healing in the negative control group treated with phenytoin and the AMONIA group were 22±3, 19.3±1.5, and 9±2 days, respectively. Therefore, there existed significant (P<0.05) differences between the study groups. The primary wound surface was approximately 100 mm2 immediately after dermal incision creation. A significant (P<0.05) decreased level of wound surface was found in the AMONIA group, compared to the control groups. In the AMONIA group, the wound was entirely generated on the 10th day of treatment; however, this value in the control group was equal to 32±1 mm2, representing the therapeutic effects of AMONIA on wound healing.
At the end of the treatment, the animals’ Mean±SEM weight was calculated as 3260±15, 3340±15, and 3380±15 g, respectively; however, no significant (P>0.05) differences were observed between the control and treatment groups. This comparison was also assessed on other treatment days; however, no significant (P>0.05) difference was found in this respect.
This technique indicated that the AMONIA was initially without Escherichia coli, Pseudomonas aeruginosa, and Staphylococcus aureus. Additionally, this substance caused a growth inhibition zone in all concentrations (20%, 40%, 60%, 80%, & 100%), representing the antimicrobial effects of AMONIA. Thus, the Mean±SEM diameter of growth inhibition zone for Staphylococcus, Escherichia coli, and Pseudomonas aeruginosa were computed as 13±2 mm2, 18±3 mm2, and 20±3 mm2, respectively. These findings indicated the inhibitory effects of this substance on common bacterial growth available in the wound site. The microbiological assessment of wound site on days 2, 5, 8, 11, 14, 17, and 20 in the control group revealed several gram-negative bacilli, negative and gram-positive cocci, and yeast. Furthermore, in the positive control group, gram-negative bacilli, gram-negative coccobacillus, and gram-positive cocci were observed.
Various macroscopic assessments were found on the daily observation of skin wounds in the treatment and control groups. As demonstrated in Figure 1, three types of skin wounds were created on the back of the animals, including negative control wound (black arrows), positive control wound (blue arrows), and AMONIA (yellow arrows). On the second day of treatment (Figure 1A), a wide-open wound with bleeding was found in the negative control group (Figure 1A, black arrow); however, the wound in the positive control represented the wound contraction (Figure 1A, blue arrow), leading to coagulation. Moreover, in the last treatment group, the AMONIA gel was applied to the skin which caused wound contraction and blood coagulation (Figure 1A, yellow arrow). On the fourth day of treatment (Figure 1B), although the coagulation was found in the negative and positive control groups (Figure 1B, black & blue arrow), in the AMONIA group (Figure 1B, yellow arrow), no coagulation was detected, leading to decreased inflammation. On the eighth day of treatment (Figure 1C), the negative control group (Figure 1C, black arrow) represented not the healing condition, compared to the AMONIA group (Figure 1C, yellow arrow); the positive control treated with phenytoin presented 15% healing rate; finally, the AMONIA treatment group (Figure 1C, yellow arrow) presented 97% dermal regeneration, compared to the controls.
.PNG)
4. Discussion
Wounds are among the superficial tissue injuries that can be healed quickly without crucial complications. However, this condition can potentially shift to a harmful pathological status that ultimately impairs tissue function. Thus, managing wound progression in the early stages of injury seems necessary to prevent further unpredictable outcomes. Applying AMONIA in acute injuries can represent effective physiological features. In this study, skin wounds were created in laboratory male rabbits and treated with AMONIA. Several factors were studied in treated wounds to evaluate the effects of this drug on wound healing in the primary stages of injury [1].
AMONIA is a natural product used for treating acute skin injuries. This substance is used immediately after acute skin wounds. AMONIA is a powder that could turn into a white gel following combination with sterile distilled water. This gel is topically applied to animals’ wounds to prevent bleeding and accelerate wound healing.
The essential properties of AMONIA belong to the presence of some natural substances, such as glycosaminoglycan, sponge extract, and magnesium. Glycosaminoglycans are long linear polysaccharides, consisting of repeating disaccharide units. Glycosaminoglycans are large molecules that quickly absorb water due to the presence of different carbohydrate classes [10]. By combining glycosaminoglycan powder with distilled water, a hydrophilic gel is immediately formed, which binds temporarily to the surrounding tissue structures. The formed gel has a high viscosity between two sterile media inside the body and a non-sterile medium outside. This agent acts as a barrier that maintains the sterility of the wound and prevents infection. Thus, in the present study, no cases of infection were found in the examined animals treated with AMONIA. Moreover, the prepared hydrogel has a high viscosity, which prevents the passage of microorganisms into the wound site; accordingly, it acts as the next characteristic in maintaining wound sterility. Sponge extract is among the natural factors in ammonia powder. This extract presents potential therapeutic properties against the activity of microorganisms, like bacteria. Sponge extract penetrates the internal environment of the bacterium and prevents proliferation; thus, it inhibits the growth of infection [11]. Magnesium is a trace element effective in multiple biological activities. A deficiency of this substance in the body causes functional defects in the cells of the immune system; therefore, magnesium plays an active role in the activity of the immune system to deal with pathogens. Magnesium plays an essential role in the respiratory bursts of immune cells in hypoxic environments. This trace element increases the rate of phagocytosis in macrophages and induces the exocytosis of immune granules. In this experimental investigation, no infection was observed in the AMONIA group. Such findings could be due to the presence of high-dose magnesium leading to the accelerated activity of immune cells available at the wound site [12].
Following exposure to blood or plasma, it was activated with an adhesive feature after approximately 1-2 minutes. Thus, the bleeding rate was reduced. The evaluated wound manifested no requiring dressing and interference with the study animals’ movements. Besides, the level of scar formation was low in the treatment sample, compared with the controls. This stage can lead to arrest in bleeding and significantly reduces the migration of immune cells to the wound site. As mentioned earlier, this type of hydrogel with antibacterial properties inhibit the presence of infection in two stages; due to its high viscosity of AMONIA hydrogel, this substance prevents the penetration of microorganism into the wound as well as exogenous microorganisms to be proliferated in wounds.
The time required for total wound healing in the AMONIA group was 13 days less than the positive control group (phenytoin group). This study revealed that AMONIA has strong antimicrobial properties, at least on Pseudomonas aeruginosa and Escherichia coli. These bacteria are the main cause of wound infection and among the most resistant gram-negative bacilli to antibiotics and chemical disinfectants. The inhibitory effects of AMONIA on coagulase-positive cocci, like Staphylococcus aureus is also of scientific interest. According to the obtained results, wound healer bioadhesive (AMONIA) accelerated wound healing, especially in the fourth to eighth treatment days. Accordingly, the wound surface and time required for recovery were reduced, and the healing rate was enhanced. Due to the mechanisms involved in skin wound healing, the AMONIA could accelerate wound healing by reducing swelling or inflammation. In other words, by modulating the inflammatory phase [13], the wound healing rate could be accelerated [14]. The prevention of wound infection and keeping the sterility of wound surface can also increase the rate of wound healing. As per the obtained data, antibiotics can control wound infections leading to wound recovery [15]. It was found that the rate of wound healing is not similar in phenytoin and AMONIA groups. Thus, the proposed phenytoin mechanisms involved in wound healing acceleration included the increased concertation of growth factors [16]; macrophages migration [17]; wound elasticity; new vascular formation; collagen synthesis, and fibroblast infiltration [18]. These potential mechanisms should be carefully evaluated.
Overall, for the first time, this study signified that the AMONIA could accelerate skin wound healing in rabbits. Additionally, the AMONIA efficacy in wound healing was found to be more effective than phenytoin 1% ointment. Thus, clinical trials in humans are recommended to find the potential mechanisms mentioned earlier, such as the effects on collagen synthesis; reduced inflammation; increased oxygen delivery; increased angiogenesis, and fibroblast filtration.
In 2007, the application of cyanoacrylate on the skin sutures of rats was accessed. This study indicated that cyanoacrylate, i.e., considered a tissue adhesive agent, is well accepted by the tissue without immunological reaction and inflammation. It was concluded that cyanoacrylate has more advantages than surgical sutures, including complexity and cost-effectiveness [19].
Hossein Nazari et al. evaluated the effects of alcoholic and aqueous extracts of oatmeal on wound healing and skin inflammation. They identified the therapeutic effects of alcoholic and aqueous extracts of oatmeal, which reduced treatment time from 11 to 4 days [20]. Conducting an experimental study between the alcoholic extract of oats and this bioremediation is strictly recommended. Various research studies have been conducted in the field of wound healing; accordingly, various healer substances have been prepared and introduced on an herbal and chemical basis. These studies include the effects of calcium, copper, and zinc ions [21, 22]; physical forces, such as radiation, electrical stimulation [23] ultrasound A waves [24], and chemicals, such as hydrocortisol [25], vitamins [26], ascorbic acid [27], phenytoin [8], saline [28], growth factors [29], and honey [26, 30].
5. Conclusion
According to the present study findings, AMONIA, through various molecular mechanisms, can inhibit tissue bleeding leading to the prevention of inflammation. This phenomenon can lead to a decreased rate of tissue damage. Additionally, this substance includes various natural substances, leading to wound healing acceleration. According to the current research findings, applying AMONIA seems useful for animal care; however, extensive efforts are required for performing human clinical trials concerning AMONIA administration.
Ethical Considerations
Compliance with ethical guidelines
All experimental procedures were conducted under the supervision of the Ethics Committee of Falavarjan University of Medical Sciences (Code: IR.FUMS.REC.1397.065).
Funding
This research did not receive any grant from funding agencies in the public, commercial, or non-profit sectors.
Authors' contributions
Conceptualization and supervision: Mohammad Hossein Asadi; Methodology: Reza Soltan Hosseini; Investigation, writing – original draft, and writing – review & editing: All authors; Data collection: Hosein Bahadoran, and Bahman Jalali Kondori; Data analysis: Bahman Jalali Kondori and Reza Soltan Hosseini.
Conflict of interest
The authors declared no conflicts of interest.
Acknowledgments
We acknowledge the Vice Chancellor for Research (Islamic Azad University, Falavarjan Branch), Dr. Hamid Fooladgar and his colleagues in the research laboratory, as well as the board members of Behzist Airik Novin Daroo (Behzist Airic Novin Daru, Iran) company; Dr. Mostafa Mohammadi, Mr. Haj Agha Mirdamadi, and Mr. Haj Agha Nasher Al-Ahkami for their support. We also acknowledge Ms. Mohadeseh Masaeli, Bahareh Sholeh, Mojgan Afshari, Maryam Ghasemi (the experts of the Islamic Azad University of Falavarjan), and Dr. Mohammad Reza Ebad (a veterinarian and the technical manager of the company) for their technical support.
- Cañedo-Dorantes L, Cañedo-Ayala M. Skin acute wound healing: A comprehensive review. International Journal of Inflammation. 2019; 2019:3706315. [DOI:10.1155/2019/3706315] [PMID] [PMCID]
- Kelmansky R, McAlvin BJ, Nyska A, Dohlman JC, Chiang HH, Hashimoto M, et al. Strong tissue glue with tunable elasticity. Acta Biomaterialia. 2017; 53:93-9. [DOI:10.1016/j.actbio.2017.02.009] [PMID]
- Cha HJ, Hwang DS, Lim S. Development of bioadhesives from marine mussels. Biotechnology Journal. 2008; 3(5):631-8. [DOI:10.1002/biot.200700258] [PMID]
- Rios LY, Gonthier MP, Rémésy C, Mila I, Lapierre C, Lazarus SA, et al. Chocolate intake increases urinary excretion of polyphenol-derived phenolic acids in healthy human subjects. The American Journal of Clinical Nutrition. 2003; 77(4):912-8. [DOI:10.1093/ajcn/77.4.912] [PMID]
- Vattem DA, Ghaedian R, Shetty K. Enhancing health benefits of berries through phenolic antioxidant enrichment: Focus on cranberry. Asia Pacific Journal of Clinical Nutrition. 2005; 14(2):120-30. [PMID]
- Arranz S, Saura-Calixto F, Shaha S, Kroon PA. High contents of nonextractable polyphenols in fruits suggest that polyphenol contents of plant foods have been underestimated. Journal of Agricultural and Food Chemistry. 2009; 57(16):7298-303. [DOI:10.1021/jf9016652] [PMID]
- Zhao H, Waite JH. Proteins in load-bearing junctions: The histidine-rich metal-binding protein of mussel byssus. Biochemistry. 2006; 45(47):14223-31. [DOI:10.1021/bi061677n] [PMID] [PMCID]
- Hasamnis A, Mohanty B, Muralikrishna, Patil S. Evaluation of wound healing effect of topical phenytoin on excisional wound in albino rats. Journal of Young Pharmacists. 2010; 2(1):59-62. [DOI:10.4103/0975-1483.62215] [PMID] [PMCID]
- Cumming BD, McElwain DLS, Upton Z. A mathematical model of wound healing and subsequent scarring. Journal of the Royal Society Interface. 2010; 7(42):19-34. [DOI:10.1098/rsif.2008.0536] [PMID] [PMCID]
- Dennis RJ, Taylor EJ, Macauley MS, Stubbs KA, Turkenburg JP, Hart SJ, et al. Structure and mechanism of a bacterial β-glucosaminidase having O-GlcNAcase activity. Nature Structural & Molecular Biology. 2006; 13(4):365-71. [DOI:10.1038/nsmb1079] [PMID]
- Beesoo R, Bhagooli R, Neergheen-Bhujun VS, Li WW, Kagansky A, Bahorun T. Antibacterial and antibiotic potentiating activities of tropical marine sponge extracts. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology. 2017; 196:81-90. [DOI:10.1016/j.cbpc.2017.04.001] [PMID]
- Dominguez LJ, Veronese N, Guerrero-Romero F, Barbagallo M. Magnesium in infectious diseases in older people. Nutrients. 2021; 13(1):180. [DOI:10.3390/nu13010180] [PMID] [PMCID]
- Yadollahpour A, Mostafa J, Samaneh R, Rezaee Z. Ultrasound therapy for wound healing: A review of current techniques and mechanisms of action. Journal of Pure and Applied Microbiology. 2014; 8(5):4071-85. https://microbiologyjournal.org/archive_mg/jmabsread.php?snoid=2223&month=&year=
- Weihs D, Gefen A, Vermolen FJ. Review on experiment-based two-and three-dimensional models for wound healing. Interface Focus. 2016; 6(5):20160038. [DOI:10.1098/rsfs.2016.0038] [PMID] [PMCID]
- Geronemus RG, Mertz PM, Eaglstein WH. Wound healing: The effects of topical antimicrobial agents. Archives of Dermatology. 1979; 115(11):1311-4. [DOI:10.1001/archderm.1979.04010110017016] [PMID]
- Dill RE, Iacopino AM. Myofibroblasts in phenytoin‐induced hyperplastic connective tissue in the rat and in human gingival overgrowth. Journal of Periodontology. 1997; 68(4):375-80. [DOI:10.1902/jop.1997.68.4.375] [PMID]
- Krzyszczyk P, Schloss R, Palmer A, Berthiaume F. The role of macrophages in acute and chronic wound healing and interventions to promote pro-wound healing phenotypes. Frontiers in Physiology. 2018; 9:419. [DOI:10.3389/fphys.2018.00419] [PMID] [PMCID]
- DaCosta ML, Regan MC, Al Sader M, Leader M, Bouchier-Hayes D. Diphenylhydantoin sodium promotes early and marked angiogenes is and results in increased collagen deposition and tensile strength in healing wounds. Surgery. 1998; 123(3):287-93. [DOI:10.1016/S0039-6060(98)70181-3] [PMID]
- de Souza SC, de Oliveira WL, de Oliveira Santos Soares DF, Briglia CH, Athanázio PR, de Cerqueira MD, et al. Comparative study of suture and Cyanoacrylates in skin closure of rats. Acta Cirúrgica Brasileira. 2007; 22(4):309-16. [DOI:10.1590/S0102-86502007000400013] [PMID]
- Nazari H, Jahanjo N, Safarieh M, Taherian M, Khaleghian A, Vafaei A, et al. [The effect of Avena sativa alcoholic and aqueous extract on the wound healing and skin inflammation (Persian)]. Studies in Medical Sciences. 2011; 22(5):467-73. http://umj.umsu.ac.ir/article-1-1118-en.html
- Borkow G. Using copper to improve the well-being of the skin. Current Chemical Biology. 2014; 8(2):89-102. [DOI:10.2174/2212796809666150227223857] [PMID] [PMCID]
- Lansdown ABG, Mirastschijski U, Stubbs N, Scanlon E, Ågren MS. Zinc in wound healing: Theoretical, experimental, and clinical aspects. Wound Repair and Regeneration. 2007; 15(1):2-16. [DOI:10.1111/j.1524-475X.2006.00179.x] [PMID]
- Kloth LC. Electrical stimulation technologies for wound healing. Advances in Wound Care. 2014; 3(2):81-90. [DOI:10.1089/wound.2013.0459] [PMID] [PMCID]
- Byl NN, McKenzie AL, West JM, Whitney JD, Hunt TK, Scheuenstuhl HA. Low-dose ultrasound effects on wound healing: A controlled study with Yucatan pigs. Archives of Physical Medicine and Rehabilitation. 1992; 73(7):656-64. [DOI:10.5555/uri:pii:000399939290132G] [PMID]
- Asboe-Hansen G. Influence of corticosteroids on connective tissue. Dermatologica. 1976; 152(Suppl 1):127-32. [DOI:10.1159/000257873] [PMID]
- Al-Waili N, Salom K, Al-Ghamdi AA. Honey for wound healing, ulcers, and burns; Data supporting its use in clinical practice. The Scientific World Journal. 2011; 11:766-87. [DOI:10.1100/tsw.2011.78] [PMID] [PMCID]
- Krámer GA, Fillios LC, Bowler EC. Ascorbic and acid treatment on early collagen production and wound healing in the guinea pig. Journal of Periodontology. 1979; 50(4):189-92. [DOI:10.1902/jop.1979.50.4.189] [PMID]
- Curtsinger LJ, Pietsch JD, Brown GL, von Fraunhofer A, Ackerman D, Polk Jr HC, et al. Reversal of Adriamycin-impaired wound healing by transforming growth factor-beta. Surgery, Gynecology & Obstetrics. 1989; 168(6):517-22. [PMID]
- Emmerson E, Campbell L, Davies FC, Ross NL, Ashcroft GS, Krust A, et al. Insulin-like growth factor-1 promotes wound healing in estrogen-deprived mice: New insights into cutaneous IGF-1R/ERα cross talk. The Journal of Investigative Dermatology. 2012; 132(12):2838-48. [DOI:10.1038/jid.2012.228] [PMID]
- Khaksari M, Rezvani ME, Sajadi MA, Soleimani A. [The effect of topically applied water extract of Rhazya stricta on cutaneous wound healing in rats (Persian)]. Koomesh. 2000; 1(3):1-10. http://koomeshjournal.semums.ac.ir/article-1-352-en.html
Type of Study: Original |
Subject:
Histology
Received: 2021/03/29 | Accepted: 2020/09/2 | Published: 2021/01/1
Received: 2021/03/29 | Accepted: 2020/09/2 | Published: 2021/01/1
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |




