Sat, Jul 12, 2025
Volume 17, Issue 2 (Summer & Autumn 2020)
ASJ 2020, 17(2): 73-82 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Sedighi P, Helli B, Sharhani A, Vatanpur A. Effects of Grape Seed Extract Supplementation on Fasting Blood Glucose, Insulin Resistance, and Lipid Profile in Women With Polycystic Ovary Syndrome. ASJ 2020; 17 (2) :73-82
URL: http://anatomyjournal.ir/article-1-267-en.html
URL: http://anatomyjournal.ir/article-1-267-en.html
1- Nutrition and Metabolic Diseases Research Center, Jundishapur University of Medical Sciences, Ahvaz, Iran, Student Research Committee, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran.
2- Nutrition and Metabolic Diseases Research Center, Jundishapur University of Medical Sciences, Ahvaz, Iran, Department of Nutrition, School of Allied Medical Sciences, Jundishapur University of Medical Sciences, Ahvaz, Iran, Department of Nutrition,Iranian Social Security Organization, Tehran, Iran.
3- Department of Epidemiology and Biostatistics School of Public Health, Jundishapur University of Medical Sciences, Ahvaz, Iran.
4- Department of Gynaecology,Iranian Social Security Organization, Tehran, Iran.
2- Nutrition and Metabolic Diseases Research Center, Jundishapur University of Medical Sciences, Ahvaz, Iran, Department of Nutrition, School of Allied Medical Sciences, Jundishapur University of Medical Sciences, Ahvaz, Iran, Department of Nutrition,Iranian Social Security Organization, Tehran, Iran.
3- Department of Epidemiology and Biostatistics School of Public Health, Jundishapur University of Medical Sciences, Ahvaz, Iran.
4- Department of Gynaecology,Iranian Social Security Organization, Tehran, Iran.
Full-Text [PDF 638 kb]
(2150 Downloads)
| Abstract (HTML) (4200 Views)
Full-Text: (2008 Views)
1. Introduction
Polycystic Ovary Syndrome (PCOS) is a widespread heterogeneous, endocrinologic, and metabolic disturbance in women. PCOS induces infertility or subfertility in females, i.e., often named hyperandrogenic anovulation or Stein-Leventhal syndrome [1, 2]. In total, 5%-10% of women are affected by PCOS in their reproductive life. In PCOS, the ovaries expand with numerous pathological cysts, containing small undeveloped follicles. The etiology of this condition remains unclear; however, it is mainly regarded as a multifactorial disturbance with genetic origins and environmental factors [3].
Women with PCOS encounter an increased risk of endometrial cancers, dyslipidemia, cardiovascular diseases, and diabetes mellitus (type II). In PCOS pathophysiology, primary defects are detected in the hypothalamic-pituitary axis, insulin secretion, insulin function, and ovarian activity [4, 5]. Based on the Rotterdam criterion (2003), the PCOS prevalence has been documented as 15.2% in Iran [6]. This disease is characterized by some special clinical features, including hyperandrogenism, anovulation, and metabolic changes (e.g., obesity, hyperinsulinemia, insulin resistance, and acanthosis nigricans) [7]. The prevalence rate of metabolic disturbances resulted from the conditions of hyperinsulinemia and dyslipidemia is crucial [3, 7, 8].
Currently, the standard PCOS therapies include lifestyle improvements and the prescription of pharmaceutical drugs, such as clomiphene citrate and metformin. Asymmetrical dimethylarginine, inflammatory, and metabolic parameters in PCOS cases were investigated in two phases; before and after metformin treatment by Heutling and colleagues (2008). In this clinical study, decreased hyperandrogenemia and ADMA levels were found; decreased ADMA levels were not correlated to Body Mass Index (BMI) improvements or metabolic parameters [9].
Chemical medicines used for PCOS treatment are associated with some adverse effects and disadvantages, such as nausea, vomiting, and stomach disorders [10]. Herbal medications have gained great attention for PCOS treatment in recent decades [11, 12]. Such medications include Ecklonia cava, Glycyrrhizaglabra, Aeglemarmelos, Bougainvillea spectabilis, Matricaria chamomile, Cinnamomumzeylanicum, Galegaofficinalis, Moringaoleifera, Nigella sativa, and hazelnut oil [13-22].
The therapeutic effects of palm pollen extract on estradiol valerate-induced PCOS rats were assessed by Jashni H.K. and associates. Decreased levels of LH, estrogen, and cystic follicle besides elevated levels of FSH, progesterone, and corpus luteum were observed after this extract administration [21]. The experimental research was applied by Anbu, J., to determine Sargassumilicifolium’s potential therapeutic effects on testosterone-induced PCOS in female Wistar rats. They found that following the administration of the ethanolic extract of Sargassumilicifolium, this plant potentially reduced the elevated glucose, cholesterol, and testosterone levels; it altered the estrus cycle to the normal state and increased the blood serum concentration of FSH, LH, estrogen, and progesterone [22].
Furthermore, the Ecklonia cava extract restored ovarian follicles components and morphologies to a healthy and physiological state. Such measures were based on regulatory effects on ovarian factors associated with the production of follicles. Eui-Ju Hong et al. explored the application effect of Ecklonia cava extract on letrozole-induced PCOS in rat models. The authors have determined that the Ecklonia cava substantially decreased PCOS symptoms. The effects of Glycyrrhizaglabra ethanol extract on letrozole-indued PCOS were assessed in Sprague Dawley rats by Hye Won Lee. In the co-administration of licorice and letrozole, a significant recovery in the FSH level and a substantial decrease in LH/FSH ratio was observed [14].
In 2018, Majid Shokoohi et al. explored a rodent model of estradiol valerate-mediated PCOS. They found the protective outcomes of Galegaofficinalis administration on metabolic as well as hormonal parameters. They also observed a significant decrease in fasting blood glucose, insulin, testosterone, LH, and FSH levels, and significant increases in serum aromatase and serum estrogen levels [19]. The effects of Matricaria chamomile (chamomile) on lipid and hormonal parameters among PCOS-affected women of reproductive age were investigated by BijanHelli and associates. Crucial reductions in total testosterone levels were observed in these cases following the oral prescription of chamomile capsule (370 mg). Moreover, no major and significant alternations were observed in LH/FSH and dehydroepiandrosterone sulfate ratios [17].
Functional Insulin Receptor (IR) presents in ovarian theca [23] and bone osteoblast [24] cells. Insulin can induce the overproduction of androgen (hyperandrogenemia) through various molecular mechanisms in theca cells; thus, the pathologic condition of hyperinsulinemia is observed in numerous patients with PCOS. Based on the signaling pathways, the IR complex could trigger the translocation of the Steroid Regulatory Element-Binding Protein 1 (SREBP-1) into the intranuclear space. Such measures are conducted through the activation of Phosphoinositide-Dependent Kinase 1 (PDK1), Phosphoinositide 3 Kinase (PI3 K), and Protein Kinase C (PKC). SREBP-1, a cellular transcription agent involving fatty acids biosynthesis, is mainly forum in cells [25].
Insulin controls the molecular process of SREBP activation and intranuclear matrix translocation. This process occurs by applying a PI3K/Akt complex and the signaling pathway for rapamycin (mTOR) mammalian target. Negatively, the Sex Hormone-Binding Globulin (SHBG) levels strictly return to the insulin levels or insulin resistance rates in PCOS-affected patients. Accordingly, insulin presents suppressive effects on SHBG. The treatment employed to enhance insulin resistance could decrease the level of androgen [26]. Increased body fat, dyslipidemia, and systemic inflammation are closely associated with IR [27]. According to the published studies, hyperglycemia can reduce the levels of antioxidants and potentially increase lipid peroxidation, leading to an irregular metabolic status. In other words, not only the PCOS is a reproductive endocrine disorder with infertility or subfertility subsequences, but it is also a sex-related metabolic disorder.
The herbal portion of grape seed is rich in bioactive compounds, such as phytochemicals, polyphenols, phenolic acids, chalcones, flavanones, and flavanols; these are used for producing cosmeceuticals and nutraceuticals. Evidence indicates the healthy and chemical preventive effects of grape-derived phytochemicals [28, 29]. In addition, certain polyphenols provide cytotoxic effects with new pharmacologically activated molecules. Most of the grape polyphenols (60%-70% of total extractable compounds) are found in seeds. There was a strong correlation between these bioactivities and a collection of highly accumulated non-polar (lipid) and semi-polar (phenolic) molecules in grape seeds, in this context [28, 30].
Based on several experimental studies, the anticancer features of molecules available in grape included proanthocyanidins, oligomeric flavonoids, phenolic acids, chalcones, flavanones, and flavanols. Grapes are rich in carbohydrate (17g/100 g), caloric content (65 kcal/100 g), and have a low glycemic index. Besides being an excellent source of manganese and potassium, these fruits also have polyphenols, B6, C, and thiamine vitamins [31, 32]. The effect of hydroalcoholic grape seed extract was investigated by Mohseni and Salbadi on PCOS in Wistar rats on primordial, secondary, preantral, and Graafian follicles, as well as corpus luteum [33]. Based on its potent anti-oxidant [33], anti-inflammatory [34], and anti-neoplastic [11] properties, GSE is commonly consumed as a dietary supplement. Procyanidins (PCs) are a member of polyphenolic compounds, containing flavan-3-ol subunits (oligomers & polymers) [34, 35].
PCs, as the most bioactive constituents of GSE, are frequently present in other food sources, such as tea, apples, and red wine [10, 11]. Proanthocyanidins present in GSE comprise oligomers or polymers of polyhydroxyflavan. The beneficial properties of proanthocyanidins are due to their conjugated and colonic metabolites [36]. The grape seed proanthocyanidins seem to have pharmacological effects. GSE, according to the antioxidant’s properties, can inhibit some illnesses’ symptoms [37].
These characteristics include anti-oxidants, anti-microbial, anti-obesity, anti-diabetic, anti-neurodegenerative, anti-osteoarthritis, anti-cancer, cardioprotective, and eye-protective properties. According to multiple studies, their chemical structure and polymerization strictly depend on the absorption and bioavailability of PCs in the gastrointestinal tract. Studies investigating GSE in combination with 5-Fluorouracil (5-FU) in normal animals are scarce [35]. The pharmacological features of PCOs, such as antioxidant and radical scavenging activities, capillary permeability, and fragility reduction, collagen destruction inhibition, and inflammation inhibition have been recorded in this respect.
In 2019, Moon et al. tested the effectiveness and safety of the oral administration of GSE proanthocyanidin in non-proliferative diabetic retinopathy patients [34]. Hemmati studied the comparative training following GSE and vitamin E administration in silicon-induced pulmonary fibrosis in rats. GSE could reduce the fibrogenic impact of silica; however, no synergistic effects were observed in this regard [37]. The effects of GSE on the lipid profile and expression of interleukin-6 in PCOS Wistar rat models were investigated by Salmabadi et al. in 2017. As a result, LDL-C, TC, IL-6 levels in the experimental groups, especially at 50 mg/kg of GSE, were decreased significantly, compared to the PCOS group; while HDL-C levels demonstrated no significant changes [38].
According to extensive experimental studies, GSE affects the metabolic status of several diseases, as an essential bioactive agent; in vitro supplementation, in vivo digestive, and the metabolic functions of GSE remain undiscovered.
2. Materials and Methods
GSE was prepared by the Shari Iran Company. The seeds were gathered from red grapes (V. Vinifera) and stored in the refrigerator for future analysis. Furthermore, a placebo was developed by the Barij Essence Company (Barij Essence, Kashan, Iran). An Enzyme-Linked Immunosorbent Assay (ELISA) kit (Monobind, CA, USA) was also used in this study. The measurement of fasting blood glucose, serum triglycerides, HDL cholesterol, and LDL cholesterol was applied through related kits (BioSystems Co, Barcelona, Spain). Nutritionist IV (First Databank, San Bruno, CA, USA) program was implemented for patent nutrient analysis.
In this double-blind, randomized, controlled clinical trial, 50 patients with PCOS (20-38 years old) and a BMI of 25-40 kg/m2 were selected from Imam Khomeini Hospital (Ahvaz City, Iran) from August 2019 to March 2020. According to the Kort and Lobo criteria for IR.10, the sample size to obtain a 95% confidence interval and 80% power was estimated as 25 cases per group. PCOS diagnosis was developed per the Rotterdam criteria (2003), requiring at least two of the three features; polycystic ovaries (detected by ultrasonography), chronic amenorrhea (or oligo-amenorrhea), and hyperandrogenism (clinical or biochemical analysis). The treatment groups were under-covered until the fundamental analyses and entire procedures were completed. Such measures included a randomized assignment, the registration of the study participants, and interventions.
All procedures were conducted by a qualified midwife at a clinic. The study participants were assigned into 2 classes; the GSE (n=25) and the placebo (n=25). GSE was administered daily (400 mg) for 8 weeks to the test group. To attain statistical validity, GSE and its placebo were concurrently rendered with the same appearance. Individuals have encoded the supplements according to the randomization list number. Due to GSE aquatic insolubility, the patients were requested to consume the supplements with each main meal for better dissolution. Moreover, a short message was sent every week to improve treatment adherence.
All study patients declared their health history. For each patient, demographic details, medical history, and anthropometric measurements (weight, height, waist & hip circumference) were also collected. Skilled workers who were blind to the identity of samples conducted all research measurements. The height of the study participants was also determined by a non-stretchable wall meter with barefoot.
Weight was assessed on a baseline and post-intervention basis (a digital scale, OMRON, Japan). The same digital scale was applied to calculate the Body Fat Percentage (BFP) through Bioelectric Impedance Analysis (BIA) between 8 AM and 10 AM when the research participants were fasting with dry and clean bare hands and legs as prescribed. We used this unit in the same setting and conditions for more precise measurements. The BMI of the study subjects was measured as the body weight (Kg) divided by the square height (m2).
For all study participants, a 24-hour recall questionnaire for a 3-day food diary (mean energy, macro- & micronutrient intake) was recorded at the beginning and end of the study (1 weekend day and 2 weekdays). The volume of consumed foods was converted to grams, and the encoded foods were analyzed for nutrients and antioxidants using a personalized nutritionist IV program (Updated for Iranian foods, First Databank, San Bruno, CA, USA).
For each research participant, a blood sample (10 mL) was gathered 8-12 hours at the beginning and end of the study. Serum aliquots were separated and stored at -70°C for biochemical studies. Blood sampling, FBS, insulin levels, and lipid profiles (10 mL) were assessed before and after conducting the interventions (10-12h overnight fasting in the morning). FBS, triglyceride, total cholesterol levels (using Pars Azmoon Kit, Iran), HDL cholesterol, LDL cholesterol (using Paadco kit, Spain), and insulin levels (using ELISA, Monobind kit, USA) were assessed biochemically. All the experiments were conducted in the laboratory of the Ahvaz Jundishapur University of Medical Sciences. Serum glucose level was quantified using normal enzymatic methods (Pars Azmun kit, Karaj, Iran). Insulin resistance was calculated using the HOMA formula index; HOMA-IR = fasting insulin (µU/mL) × fasting blood glucose (mg/dL)/405.
Standard Pars Azmun kit (Karaj, Iran) enzyme approaches were employed to assess Total serum Cholesterol (TC), Triacylglycerol (TG), and High-density Lipoprotein (HDL-C). The Friedewald index calculated the concentration of LDL cholesterol; LDL-C = TC − (HDL-C + TG/5)×31.
All measurements (including anthropometric indices, dietary intakes, blood sampling & biochemical assessments) were applied again at the end of the intervention procedures in the treatment and placebo groups.
The obtained data were analyzed using SPSS. Mean±SD or frequency(percentage) was used for reporting the collected data. To investigate the distribution of the variables, the Kolmogorov–Smirnov test was used. Independent Samples t-test and Paired Samples t-test was also used for quantitative data assessments, respectively for between-group and within-group comparisons. Based on a previous study, the sample size [23] was calculated considering type one error (α) of 0.05 and type 2 error (β) of 0.20 (the power of 80%), and the expectations value of 32 as the difference in the mean(d) value of TG levels as the critical variable. We reached 50 patients, and the final sample size was determined as 25 patients per group.
3. Results
Table 1 lists the demographic data of PCOS cases. At the beginning of the study, the general characteristics of the study subjects were collected (Table 1). There were no significant differences in weight, BMI, and other body measures between the study groups during 8 weeks of intervention. Considerable weight and BMI differences were observed in the GSE group, compared to their baseline values. However, there were no improvements in weight and BMI in the control group.
.PNG)
Table 2 indicates the differences between the study variables. The Analysis of Covariance (ANCOVA) findings revealed statistically significant variations between two weight groups adjusted for energy consumption and baseline value at the end of the study. BMI alteration was not significant between the two groups at the end of the study. Additionally, the total consumption of energy and macronutrients was not significantly changed during the study. Table 2 demonstrates changes in the study variables between the treatment and placebo groups. Moreover, BMI changes were not significant in the treatment group, compared to the placebo group. Total energy intake and macronutrient consumption presented no significant differences between the study groups.
Dietary energy intake, macronutrients, and micronutrients at the baseline and 8-week of GSE supplementation are presented in Table 3.
Furthermore, Table 1 contains the characteristics of the study subjects concerning daily micronutrients and macronutrients at the beginning of the study. There were no major variations in the consumption of macronutrients and micronutrients between the studied groups.
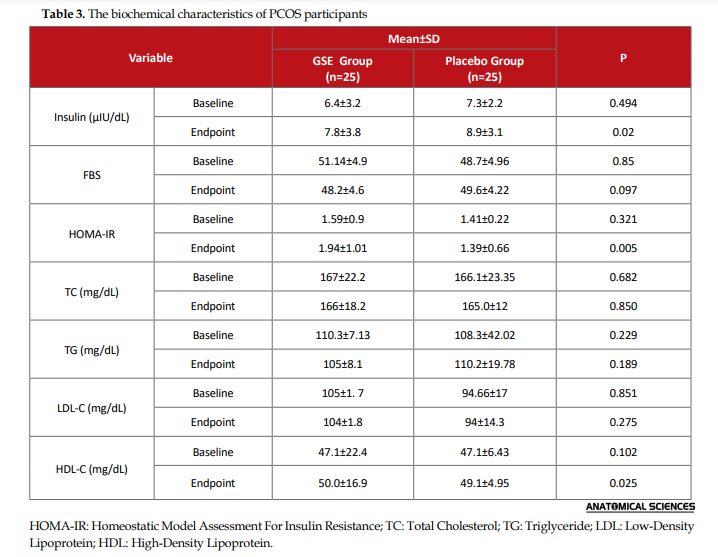
In Table 2, the alteration of biochemical variables following the intervention is demonstrated between the study groups. ANCOVA data revealed statistically important variations in specific biochemical parameters. ANCOVA results suggested statistically significant differences in serum insulin (P=0.005) and HOMA (P=0.02) levels.
.PNG)
4. Discussion
Due to antidiabetic features, the seeds of the grapes are among the crucial species with most applications in traditional medicines. Phenolics, polyphenols, vitamin E, oligomeric procyanidin, and proanthocyanidins are the main components in GSE. Proanthocyanidins supplementation has major bioactive characteristics.
GSE provided no significant alteration in the lipid profiles of the investigated women with PCOS. Hyperlipidemia plays a key role in numerous conditions, such as obesity, diabetes, inflammation, and atherosclerosis. Hyperlipidemia plays a vital role in various diseases’ pathophysiology, such as obesity, diabetes, inflammation, and atherosclerosis. This pathologic condition is more pronounced in women with PCOS. Decreased HDL-C, as well as increased triglycerides and LDL-C concentrations, are the most prevalent lipid disturbances in subjects with PCOS.
GSE is a free radical scavenging agent, i.e., involved in lipid homeostasis with reduced risk of atherosclerosis and inflammation in blood plasma, cells, or tissues. The effect of GSE on glucose homeostasis parameters is accounted for by different mechanisms, including the activation of peroxisome proliferator-activated receptor-gamma. This result is similar to some animal studies, such as Sprague Dawley rats with alloxan obese diabetic induction and high fat dietary mice. The sample size of our trial was limited; thus, it could be difficult to detect small improvements following GSE treatment.
Our study was confined to selected metabolic parameters; accordingly, we were unable to establish other biomarkers, such as endogenous antioxidants (SOD), catalase, and glutathione levels. It would be useful to research the impact of GSE on reproductive hormones, including androgen levels and SHBG. The quantification of serum or plasma GSE levels is also recommended. Furthermore, more extensive scale studies concerning various doses and treatment duration are warranted to support these findings.
5. Conclusion
Generally, the current trial suggested that the short-term use of GSE supplementation could be beneficial in women with PCOS. Such a measure could improve serum insulin and fasting blood glucose status, insulin resistance, and lipid profile in them. In the GSE treatment group, the serum HOMA-IR and FBS decreased significantly, compared to fundamental values (P=0.005 & P=0.02, respectively). Serum insulin was increased in the GSE group, without significant changes, compared to the baseline values. In the GSE group, triglyceride and body weight index were decreased, in comparison with the baseline rates. Serum TC and LDL-C levels presented no substantial changes in either group. Short-term GSE supplementation provided some positive effects on the studied women with PCOS in terms of metabolic risk factors, like HOMA-IR, which could be useful for PCOS regulation. Further investigations are suggested to create a clear link between GSE and glycemia regulation.
Ethical Considerations
Compliance with ethical guidelines
This study was approved by the Ethics Committee of Jundishapour university of medical sciences.
Funding
The present study financially supported by the Nutrition and Metabolic Disease Research Center of Ahvaz Jundishapur University of Medical Sciences (Grant Code: NRC-9604).
Authors' contributions
All authors contributed equally in preparing all parts of the research.
Conflict of interest
Supervision: Bijan Helli. Methodology: Pegah Sedighi. Writing – original draft: Pegah Sedighi. Review & editing: All authors.
Refrences:
Polycystic Ovary Syndrome (PCOS) is a widespread heterogeneous, endocrinologic, and metabolic disturbance in women. PCOS induces infertility or subfertility in females, i.e., often named hyperandrogenic anovulation or Stein-Leventhal syndrome [1, 2]. In total, 5%-10% of women are affected by PCOS in their reproductive life. In PCOS, the ovaries expand with numerous pathological cysts, containing small undeveloped follicles. The etiology of this condition remains unclear; however, it is mainly regarded as a multifactorial disturbance with genetic origins and environmental factors [3].
Women with PCOS encounter an increased risk of endometrial cancers, dyslipidemia, cardiovascular diseases, and diabetes mellitus (type II). In PCOS pathophysiology, primary defects are detected in the hypothalamic-pituitary axis, insulin secretion, insulin function, and ovarian activity [4, 5]. Based on the Rotterdam criterion (2003), the PCOS prevalence has been documented as 15.2% in Iran [6]. This disease is characterized by some special clinical features, including hyperandrogenism, anovulation, and metabolic changes (e.g., obesity, hyperinsulinemia, insulin resistance, and acanthosis nigricans) [7]. The prevalence rate of metabolic disturbances resulted from the conditions of hyperinsulinemia and dyslipidemia is crucial [3, 7, 8].
Currently, the standard PCOS therapies include lifestyle improvements and the prescription of pharmaceutical drugs, such as clomiphene citrate and metformin. Asymmetrical dimethylarginine, inflammatory, and metabolic parameters in PCOS cases were investigated in two phases; before and after metformin treatment by Heutling and colleagues (2008). In this clinical study, decreased hyperandrogenemia and ADMA levels were found; decreased ADMA levels were not correlated to Body Mass Index (BMI) improvements or metabolic parameters [9].
Chemical medicines used for PCOS treatment are associated with some adverse effects and disadvantages, such as nausea, vomiting, and stomach disorders [10]. Herbal medications have gained great attention for PCOS treatment in recent decades [11, 12]. Such medications include Ecklonia cava, Glycyrrhizaglabra, Aeglemarmelos, Bougainvillea spectabilis, Matricaria chamomile, Cinnamomumzeylanicum, Galegaofficinalis, Moringaoleifera, Nigella sativa, and hazelnut oil [13-22].
The therapeutic effects of palm pollen extract on estradiol valerate-induced PCOS rats were assessed by Jashni H.K. and associates. Decreased levels of LH, estrogen, and cystic follicle besides elevated levels of FSH, progesterone, and corpus luteum were observed after this extract administration [21]. The experimental research was applied by Anbu, J., to determine Sargassumilicifolium’s potential therapeutic effects on testosterone-induced PCOS in female Wistar rats. They found that following the administration of the ethanolic extract of Sargassumilicifolium, this plant potentially reduced the elevated glucose, cholesterol, and testosterone levels; it altered the estrus cycle to the normal state and increased the blood serum concentration of FSH, LH, estrogen, and progesterone [22].
Furthermore, the Ecklonia cava extract restored ovarian follicles components and morphologies to a healthy and physiological state. Such measures were based on regulatory effects on ovarian factors associated with the production of follicles. Eui-Ju Hong et al. explored the application effect of Ecklonia cava extract on letrozole-induced PCOS in rat models. The authors have determined that the Ecklonia cava substantially decreased PCOS symptoms. The effects of Glycyrrhizaglabra ethanol extract on letrozole-indued PCOS were assessed in Sprague Dawley rats by Hye Won Lee. In the co-administration of licorice and letrozole, a significant recovery in the FSH level and a substantial decrease in LH/FSH ratio was observed [14].
In 2018, Majid Shokoohi et al. explored a rodent model of estradiol valerate-mediated PCOS. They found the protective outcomes of Galegaofficinalis administration on metabolic as well as hormonal parameters. They also observed a significant decrease in fasting blood glucose, insulin, testosterone, LH, and FSH levels, and significant increases in serum aromatase and serum estrogen levels [19]. The effects of Matricaria chamomile (chamomile) on lipid and hormonal parameters among PCOS-affected women of reproductive age were investigated by BijanHelli and associates. Crucial reductions in total testosterone levels were observed in these cases following the oral prescription of chamomile capsule (370 mg). Moreover, no major and significant alternations were observed in LH/FSH and dehydroepiandrosterone sulfate ratios [17].
Functional Insulin Receptor (IR) presents in ovarian theca [23] and bone osteoblast [24] cells. Insulin can induce the overproduction of androgen (hyperandrogenemia) through various molecular mechanisms in theca cells; thus, the pathologic condition of hyperinsulinemia is observed in numerous patients with PCOS. Based on the signaling pathways, the IR complex could trigger the translocation of the Steroid Regulatory Element-Binding Protein 1 (SREBP-1) into the intranuclear space. Such measures are conducted through the activation of Phosphoinositide-Dependent Kinase 1 (PDK1), Phosphoinositide 3 Kinase (PI3 K), and Protein Kinase C (PKC). SREBP-1, a cellular transcription agent involving fatty acids biosynthesis, is mainly forum in cells [25].
Insulin controls the molecular process of SREBP activation and intranuclear matrix translocation. This process occurs by applying a PI3K/Akt complex and the signaling pathway for rapamycin (mTOR) mammalian target. Negatively, the Sex Hormone-Binding Globulin (SHBG) levels strictly return to the insulin levels or insulin resistance rates in PCOS-affected patients. Accordingly, insulin presents suppressive effects on SHBG. The treatment employed to enhance insulin resistance could decrease the level of androgen [26]. Increased body fat, dyslipidemia, and systemic inflammation are closely associated with IR [27]. According to the published studies, hyperglycemia can reduce the levels of antioxidants and potentially increase lipid peroxidation, leading to an irregular metabolic status. In other words, not only the PCOS is a reproductive endocrine disorder with infertility or subfertility subsequences, but it is also a sex-related metabolic disorder.
The herbal portion of grape seed is rich in bioactive compounds, such as phytochemicals, polyphenols, phenolic acids, chalcones, flavanones, and flavanols; these are used for producing cosmeceuticals and nutraceuticals. Evidence indicates the healthy and chemical preventive effects of grape-derived phytochemicals [28, 29]. In addition, certain polyphenols provide cytotoxic effects with new pharmacologically activated molecules. Most of the grape polyphenols (60%-70% of total extractable compounds) are found in seeds. There was a strong correlation between these bioactivities and a collection of highly accumulated non-polar (lipid) and semi-polar (phenolic) molecules in grape seeds, in this context [28, 30].
Based on several experimental studies, the anticancer features of molecules available in grape included proanthocyanidins, oligomeric flavonoids, phenolic acids, chalcones, flavanones, and flavanols. Grapes are rich in carbohydrate (17g/100 g), caloric content (65 kcal/100 g), and have a low glycemic index. Besides being an excellent source of manganese and potassium, these fruits also have polyphenols, B6, C, and thiamine vitamins [31, 32]. The effect of hydroalcoholic grape seed extract was investigated by Mohseni and Salbadi on PCOS in Wistar rats on primordial, secondary, preantral, and Graafian follicles, as well as corpus luteum [33]. Based on its potent anti-oxidant [33], anti-inflammatory [34], and anti-neoplastic [11] properties, GSE is commonly consumed as a dietary supplement. Procyanidins (PCs) are a member of polyphenolic compounds, containing flavan-3-ol subunits (oligomers & polymers) [34, 35].
PCs, as the most bioactive constituents of GSE, are frequently present in other food sources, such as tea, apples, and red wine [10, 11]. Proanthocyanidins present in GSE comprise oligomers or polymers of polyhydroxyflavan. The beneficial properties of proanthocyanidins are due to their conjugated and colonic metabolites [36]. The grape seed proanthocyanidins seem to have pharmacological effects. GSE, according to the antioxidant’s properties, can inhibit some illnesses’ symptoms [37].
These characteristics include anti-oxidants, anti-microbial, anti-obesity, anti-diabetic, anti-neurodegenerative, anti-osteoarthritis, anti-cancer, cardioprotective, and eye-protective properties. According to multiple studies, their chemical structure and polymerization strictly depend on the absorption and bioavailability of PCs in the gastrointestinal tract. Studies investigating GSE in combination with 5-Fluorouracil (5-FU) in normal animals are scarce [35]. The pharmacological features of PCOs, such as antioxidant and radical scavenging activities, capillary permeability, and fragility reduction, collagen destruction inhibition, and inflammation inhibition have been recorded in this respect.
In 2019, Moon et al. tested the effectiveness and safety of the oral administration of GSE proanthocyanidin in non-proliferative diabetic retinopathy patients [34]. Hemmati studied the comparative training following GSE and vitamin E administration in silicon-induced pulmonary fibrosis in rats. GSE could reduce the fibrogenic impact of silica; however, no synergistic effects were observed in this regard [37]. The effects of GSE on the lipid profile and expression of interleukin-6 in PCOS Wistar rat models were investigated by Salmabadi et al. in 2017. As a result, LDL-C, TC, IL-6 levels in the experimental groups, especially at 50 mg/kg of GSE, were decreased significantly, compared to the PCOS group; while HDL-C levels demonstrated no significant changes [38].
According to extensive experimental studies, GSE affects the metabolic status of several diseases, as an essential bioactive agent; in vitro supplementation, in vivo digestive, and the metabolic functions of GSE remain undiscovered.
2. Materials and Methods
GSE was prepared by the Shari Iran Company. The seeds were gathered from red grapes (V. Vinifera) and stored in the refrigerator for future analysis. Furthermore, a placebo was developed by the Barij Essence Company (Barij Essence, Kashan, Iran). An Enzyme-Linked Immunosorbent Assay (ELISA) kit (Monobind, CA, USA) was also used in this study. The measurement of fasting blood glucose, serum triglycerides, HDL cholesterol, and LDL cholesterol was applied through related kits (BioSystems Co, Barcelona, Spain). Nutritionist IV (First Databank, San Bruno, CA, USA) program was implemented for patent nutrient analysis.
In this double-blind, randomized, controlled clinical trial, 50 patients with PCOS (20-38 years old) and a BMI of 25-40 kg/m2 were selected from Imam Khomeini Hospital (Ahvaz City, Iran) from August 2019 to March 2020. According to the Kort and Lobo criteria for IR.10, the sample size to obtain a 95% confidence interval and 80% power was estimated as 25 cases per group. PCOS diagnosis was developed per the Rotterdam criteria (2003), requiring at least two of the three features; polycystic ovaries (detected by ultrasonography), chronic amenorrhea (or oligo-amenorrhea), and hyperandrogenism (clinical or biochemical analysis). The treatment groups were under-covered until the fundamental analyses and entire procedures were completed. Such measures included a randomized assignment, the registration of the study participants, and interventions.
All procedures were conducted by a qualified midwife at a clinic. The study participants were assigned into 2 classes; the GSE (n=25) and the placebo (n=25). GSE was administered daily (400 mg) for 8 weeks to the test group. To attain statistical validity, GSE and its placebo were concurrently rendered with the same appearance. Individuals have encoded the supplements according to the randomization list number. Due to GSE aquatic insolubility, the patients were requested to consume the supplements with each main meal for better dissolution. Moreover, a short message was sent every week to improve treatment adherence.
All study patients declared their health history. For each patient, demographic details, medical history, and anthropometric measurements (weight, height, waist & hip circumference) were also collected. Skilled workers who were blind to the identity of samples conducted all research measurements. The height of the study participants was also determined by a non-stretchable wall meter with barefoot.
Weight was assessed on a baseline and post-intervention basis (a digital scale, OMRON, Japan). The same digital scale was applied to calculate the Body Fat Percentage (BFP) through Bioelectric Impedance Analysis (BIA) between 8 AM and 10 AM when the research participants were fasting with dry and clean bare hands and legs as prescribed. We used this unit in the same setting and conditions for more precise measurements. The BMI of the study subjects was measured as the body weight (Kg) divided by the square height (m2).
For all study participants, a 24-hour recall questionnaire for a 3-day food diary (mean energy, macro- & micronutrient intake) was recorded at the beginning and end of the study (1 weekend day and 2 weekdays). The volume of consumed foods was converted to grams, and the encoded foods were analyzed for nutrients and antioxidants using a personalized nutritionist IV program (Updated for Iranian foods, First Databank, San Bruno, CA, USA).
For each research participant, a blood sample (10 mL) was gathered 8-12 hours at the beginning and end of the study. Serum aliquots were separated and stored at -70°C for biochemical studies. Blood sampling, FBS, insulin levels, and lipid profiles (10 mL) were assessed before and after conducting the interventions (10-12h overnight fasting in the morning). FBS, triglyceride, total cholesterol levels (using Pars Azmoon Kit, Iran), HDL cholesterol, LDL cholesterol (using Paadco kit, Spain), and insulin levels (using ELISA, Monobind kit, USA) were assessed biochemically. All the experiments were conducted in the laboratory of the Ahvaz Jundishapur University of Medical Sciences. Serum glucose level was quantified using normal enzymatic methods (Pars Azmun kit, Karaj, Iran). Insulin resistance was calculated using the HOMA formula index; HOMA-IR = fasting insulin (µU/mL) × fasting blood glucose (mg/dL)/405.
Standard Pars Azmun kit (Karaj, Iran) enzyme approaches were employed to assess Total serum Cholesterol (TC), Triacylglycerol (TG), and High-density Lipoprotein (HDL-C). The Friedewald index calculated the concentration of LDL cholesterol; LDL-C = TC − (HDL-C + TG/5)×31.
All measurements (including anthropometric indices, dietary intakes, blood sampling & biochemical assessments) were applied again at the end of the intervention procedures in the treatment and placebo groups.
The obtained data were analyzed using SPSS. Mean±SD or frequency(percentage) was used for reporting the collected data. To investigate the distribution of the variables, the Kolmogorov–Smirnov test was used. Independent Samples t-test and Paired Samples t-test was also used for quantitative data assessments, respectively for between-group and within-group comparisons. Based on a previous study, the sample size [23] was calculated considering type one error (α) of 0.05 and type 2 error (β) of 0.20 (the power of 80%), and the expectations value of 32 as the difference in the mean(d) value of TG levels as the critical variable. We reached 50 patients, and the final sample size was determined as 25 patients per group.
3. Results
Table 1 lists the demographic data of PCOS cases. At the beginning of the study, the general characteristics of the study subjects were collected (Table 1). There were no significant differences in weight, BMI, and other body measures between the study groups during 8 weeks of intervention. Considerable weight and BMI differences were observed in the GSE group, compared to their baseline values. However, there were no improvements in weight and BMI in the control group.
.PNG)
Table 2 indicates the differences between the study variables. The Analysis of Covariance (ANCOVA) findings revealed statistically significant variations between two weight groups adjusted for energy consumption and baseline value at the end of the study. BMI alteration was not significant between the two groups at the end of the study. Additionally, the total consumption of energy and macronutrients was not significantly changed during the study. Table 2 demonstrates changes in the study variables between the treatment and placebo groups. Moreover, BMI changes were not significant in the treatment group, compared to the placebo group. Total energy intake and macronutrient consumption presented no significant differences between the study groups.
Dietary energy intake, macronutrients, and micronutrients at the baseline and 8-week of GSE supplementation are presented in Table 3.

Furthermore, Table 1 contains the characteristics of the study subjects concerning daily micronutrients and macronutrients at the beginning of the study. There were no major variations in the consumption of macronutrients and micronutrients between the studied groups.
In Table 2, the alteration of biochemical variables following the intervention is demonstrated between the study groups. ANCOVA data revealed statistically important variations in specific biochemical parameters. ANCOVA results suggested statistically significant differences in serum insulin (P=0.005) and HOMA (P=0.02) levels.
.PNG)
4. Discussion
Due to antidiabetic features, the seeds of the grapes are among the crucial species with most applications in traditional medicines. Phenolics, polyphenols, vitamin E, oligomeric procyanidin, and proanthocyanidins are the main components in GSE. Proanthocyanidins supplementation has major bioactive characteristics.
GSE provided no significant alteration in the lipid profiles of the investigated women with PCOS. Hyperlipidemia plays a key role in numerous conditions, such as obesity, diabetes, inflammation, and atherosclerosis. Hyperlipidemia plays a vital role in various diseases’ pathophysiology, such as obesity, diabetes, inflammation, and atherosclerosis. This pathologic condition is more pronounced in women with PCOS. Decreased HDL-C, as well as increased triglycerides and LDL-C concentrations, are the most prevalent lipid disturbances in subjects with PCOS.
GSE is a free radical scavenging agent, i.e., involved in lipid homeostasis with reduced risk of atherosclerosis and inflammation in blood plasma, cells, or tissues. The effect of GSE on glucose homeostasis parameters is accounted for by different mechanisms, including the activation of peroxisome proliferator-activated receptor-gamma. This result is similar to some animal studies, such as Sprague Dawley rats with alloxan obese diabetic induction and high fat dietary mice. The sample size of our trial was limited; thus, it could be difficult to detect small improvements following GSE treatment.
Our study was confined to selected metabolic parameters; accordingly, we were unable to establish other biomarkers, such as endogenous antioxidants (SOD), catalase, and glutathione levels. It would be useful to research the impact of GSE on reproductive hormones, including androgen levels and SHBG. The quantification of serum or plasma GSE levels is also recommended. Furthermore, more extensive scale studies concerning various doses and treatment duration are warranted to support these findings.
5. Conclusion
Generally, the current trial suggested that the short-term use of GSE supplementation could be beneficial in women with PCOS. Such a measure could improve serum insulin and fasting blood glucose status, insulin resistance, and lipid profile in them. In the GSE treatment group, the serum HOMA-IR and FBS decreased significantly, compared to fundamental values (P=0.005 & P=0.02, respectively). Serum insulin was increased in the GSE group, without significant changes, compared to the baseline values. In the GSE group, triglyceride and body weight index were decreased, in comparison with the baseline rates. Serum TC and LDL-C levels presented no substantial changes in either group. Short-term GSE supplementation provided some positive effects on the studied women with PCOS in terms of metabolic risk factors, like HOMA-IR, which could be useful for PCOS regulation. Further investigations are suggested to create a clear link between GSE and glycemia regulation.
Ethical Considerations
Compliance with ethical guidelines
This study was approved by the Ethics Committee of Jundishapour university of medical sciences.
Funding
The present study financially supported by the Nutrition and Metabolic Disease Research Center of Ahvaz Jundishapur University of Medical Sciences (Grant Code: NRC-9604).
Authors' contributions
All authors contributed equally in preparing all parts of the research.
Conflict of interest
Supervision: Bijan Helli. Methodology: Pegah Sedighi. Writing – original draft: Pegah Sedighi. Review & editing: All authors.
Refrences:
- Kamble AV, Dhamane SP, Kulkarni AS, Potnis VV. Review on effects of herbal extract for the treatment of polycystic ovarian syndrome (PCOS). Plant Archives. 2020; 20(1):1189-95. http://plantarchives.org/20-1/1189-1195%20(5820).pdf
- Krishnan A, Muthusami S. Hormonal alterations in PCOS and its influence on bone metabolism. Journal of Endocrinology. 2017; 232(2):R99-113. [DOI:10.1530/JOE-16-0405] [PMID]
- Amini L, Mojab F, Jahanfar S, Sepidarkish M, Raoofi Z, Maleki-Hajiagha A. Efficacy of Salvia officinalis extract on the prevention of insulin resistance in euglycemic patients with polycystic ovary syndrome: A double-blinded placebo-controlled clinical trial. Complementary Therapies in Medicine. 2020; 48:102245. [DOI:10.1016/j.ctim.2019.102245] [PMID]
- Paradisi G, Steinberg HO, Hempfling A, Cronin J, Hook G, Shepard MK, Baron AD. Polycystic ovary syndrome is associated with endothelial dysfunction. Circulation. 2001; 103(10):1410-5. [DOI:10.1161/01.CIR.103.10.1410] [PMID]
- Fazelian S, Rouhani MH, Bank SS, Amani R. Chromium supplementation and polycystic ovary syndrome: A systematic review and meta-analysis. Journal of Trace Elements in Medicine and Biology. 2017; 42:92-6. [DOI:10.1016/j.jtemb.2017.04.008] [PMID]
- Azziz R. Controversy in clinical endocrinology: Diagnosis of polycystic ovarian syndrome: The Rotterdam criteria are premature. The Journal of Clinical Endocrinology & Metabolism. 2006; 91(3):781-5.[DOI:10.1210/jc.2005-2153] [PMID]
- Aroda V, Ciaraldi TP, Chang S-A, Dahan MH, Chang RJ, Henry RR. Circulating and cellular adiponectin in polycystic ovary syndrome: Relationship to glucose tolerance and insulin action. Fertility and Sterility. 2008; 89(5):1200-8. [DOI:10.1016/j.fertnstert.2007.04.046] [PMID] [PMCID]
- Ngo DTM, Chan WP, Rajendran S, Heresztyn T, Amarasekera A, Sverdlov AL, et al. Determinants of insulin responsiveness in young women: Impact of polycystic ovarian syndrome, nitric oxide, and vitamin D. Nitric Oxide. 2011; 25(3):326-30. [DOI:10.1016/j.niox.2011.06.005] [PMID]
- Ek I, Arner P, Rydén M, Holm C, Thörne A, Hoffstedt J, et al. A unique defect in the regulation of visceral fat cell lipolysis in the polycystic ovary syndrome as an early link to insulin resistance. Diabetes. 2002; 51(2):484-92.[DOI:10.2337/diabetes.51.2.484] [PMID]
- Arentz S, Abbott JA, Smith CA, Bensoussan A. Herbal medicine for the management of polycystic ovary syndrome (PCOS) and associated oligo/amenorrhoea and hyperandrogenism; a review of the laboratory evidence for effects with corroborative clinical findings. BMC Complementary and Alternative Medicine. 2014; 14:511. [DOI:10.1186/1472-6882-14-511] [PMID] [PMCID]
- Mao JT, Xue B, Smoake J, Lu QY, Park H, Henning SM, et al. MicroRNA-19a/b mediates grape seed procyanidin extract-induced anti-neoplastic effects against lung cancer. The Journal of Nutritional Biochemistry. 2016; 34:118-25. [DOI:10.1016/j.jnutbio.2016.05.003] [PMID]
- Mohammadshahi M, Zakizadeh E, Ahmadi-Angali K, Ravanbakhsh M, Helli B. The synergic effects of alpha-lipoic acid supplementation and electrical isotonic contraction on anthropometric measurements and the serum levels of VEGF, NO, sirtuin-1, and PGC1-α in obese people undergoing a weight loss diet. Archives of Physiology and Biochemistry. 2020:1-7. [DOI:10.1080/13813455.2020.1762660] [PMID]
- Yang H, Lee SY, Lee SR, Pyun BJ, Kim HJ, Lee YH, et al. Therapeutic effect of Ecklonia cava extract in letrozole-induced polycystic ovary syndrome rats. Frontiers in Pharmacology. 2018; 9:1325. [DOI:10.3389/fphar.2018.01325] [PMID] [PMCID]
- Yang H, Kim HJ, Pyun BJ, Lee HW. Licorice ethanol extract improves symptoms of polycytic ovary syndrome in Letrozole-induced female rats. Integrative Medicine Research. 2018; 7(3):264-70. [DOI:10.1016/j.imr.2018.05.003] [PMID] [PMCID]
- Dhivya C, Dhanalakshmi S, Chitra V, Hawari S. Alleviation of polycystic ovarian syndrome by hydroalcoholic leaf extract of Aegle marmelos (L). Correa in letrozole-induced rat model. Drug Invention Today. 2018; 10(7):1246-50. https://jprsolutions.info/files/final-file-5b424ac430e304.28323202.pdf
- Badawi AM, Ebrahim NA, Ahmed SB, Hassan AA, Khaled DM. The possible protective effect of Bougainvillea spectabilis leaves extract on estradiol valerate-induced polycystic ovary syndrome in rats (biochemical and histological study). European Journal of Anatomy. 2018; 22(6):461-9. http://www.eurjanat.com/data/pdf/eja.180205ne.pdf
- Haidari F, Homayouni F, Helli B, Haghighizadeh MH, Farahmandpour F. Effect of chlorella supplementation on systematic symptoms and serum levels of prostaglandins, inflammatory and oxidative markers in women with primary dysmenorrhea. European Journal of Obstetrics & Gynecology and Reproductive Biology. 2018; 229:185-9. [DOI: 10.1016/j.ejogrb.2018.08.578] [PMID]
- Borzoei A, Rafraf M, Niromanesh S, Farzadi L, Narimani F, Doostan F. Effects of cinnamon supplementation on antioxidant status and serum lipids in women with polycystic ovary syndrome. Journal of Traditional and Complementary Medicine. 2018; 8(1):128-33. [DOI:10.1016/j.jtcme.2017.04.008] [PMID] [PMCID]
- Abtahi-Eivari SH, Moghimian M, Soltani M, Shoorei H, Asghari R, Hajizadeh H, et al. The effect of Galega officinalis on hormonal and metabolic profile in a rat model of polycystic ovary syndrome. International Journal of Womens Health and Reproduction Sciences. 2018; 6(3):276-82. [DOI:10.15296/ijwhr.2018.46]
- Amelia D, Santoso B, Purwanto B, Miftahussurur M, Joewono HT. Effects of Moringa oleifera on insulin levels and folliculogenesis in polycystic ovary syndrome model with insulin resistance. Immunology, Endocrine & Metabolic Agents in Medicinal Chemistry. 2018; 18(1):22-30. [DOI:10.2174/1871522218666180426100754] [PMID] [PMCID]
- Karimi Jashni H, Kargar Jahromi H, Bagheri Z. The effect of palm pollen extract on polycystic ovary syndrome (POS) in rats. International Journal of Medical Research & Health Science. 2016; 5(5):317-21. http://eprints.jums.ac.ir/129/1/the-effect-of-palm-pollen-extract-on-polycystic-ovary-syndrome-pos-in-rats.pdf
- Anbu J, Sukanya K, Santhosh Kumar S, Ramya PSR, Nandihalli VB. Effect of Sargassum ilicifolium on ovogenesis in polycystic ovary syndrome-induced rats. Asian Journal of Pharmaceutical and Clinical Research. 2016; 9(6):127-3. [DOI:10.22159/ajpcr.2016.v9i6.13837]
- Nestler JE, Jakubowicz DJ, Evans WS, Pasquali R. Effects of metformin on spontaneous and clomiphene-induced ovulation in the polycystic ovary syndrome. New England Journal of Medicine. 1998; 338(26):1876-80. [DOI:10.1056/NEJM199806253382603] [PMID]
- Wang L, Zhong Y, Ding Y, Shi X, Huang J, Zhu F. Elevated serum chemerin in Chinese women with hyperandrogenic PCOS. Gynecological Endocrinology. 2014; 30(10):746-50. [DOI:10.3109/09513590.2014.928687] [PMID]
- Shao W, Espenshade PJ. Expanding roles for SREBP in metabolism. Cell Metabolism. 2012; 16(4):414-9.[DOI:10.1016/j.cmet.2012.09.002] [PMID] [PMCID]
- Baptiste CG, Battista M-C, Trottier A, Baillargeon J-P. Insulin and hyperandrogenism in women with polycystic ovary syndrome. The Journal of Steroid Biochemistry and Molecular Biology. 2010; 122(1-3):42-52. [DOI:10.1016/j.jsbmb.2009.12.010] [PMID] [PMCID]
- Sohaei S, Amani R, Tarrahi MJ, Ghasemi-Tehrani H. The effects of curcumin supplementation on glycemic status, lipid profile and hs-CRP levels in overweight/obese women with polycystic ovary syndrome: A randomized, double-blind, placebo-controlled clinical trial. Complementary Therapies in Medicine. 2019; 47:102201.[DOI:10.1016/j.ctim.2019.102201] [PMID]
- Moreno DA, Ilic N, Poulev A, Brasaemle DL, Fried SK, Raskin I. Inhibitory effects of grape seed extract on lipases. Nutrition. 2003; 19(10):876-9. [DOI:10.1016/S0899-9007(03)00167-9] [PMID]
- Özden FO, Sakallioğlu EE, Sakallioğlu U, Ayas B, Erişgin Z. Effects of grape seed extract on periodontal disease: an experimental study in rats. Journal of Applied Oral Science. 2017; 25(2):121-9. [DOI:10.1590/1678-77572016-0298] [PMID] [PMCID]
- Mohseni Kouchesfahani H, Parivar K, Salmabadi Z. [Effect of hydroalcoholic grape seed extract (Vitis vinifera L.) on polycystic ovarian syndrome in female Wistar rat (Persian)]. Journal of Cell & Tissue. 2015; 6(2):153-64. http://jct.araku.ac.ir/article_14086.html?lang=en
- Sano A, Yamakoshi J, Tokutake S, Tobe K, Kubota Y, Kikuchi M. Procyanidin B1 is detected in human serum after intake of proanthocyanidin-rich grape seed extract. Bioscience, Biotechnology, and Biochemistry. 2003; 67(5):1140-3. [DOI:10.1271/bbb.67.1140] [PMID]
- Barbe A, Ramé C, Mellouk N, Estienne A, Bongrani A, Brossaud A, et al. Effects of grape seed extract and proanthocyanidin B2 on in vitro proliferation, viability, steroidogenesis, oxidative stress, and cell signaling in human granulosa cells. International Journal of Molecular Sciences. 2019; 20(17):4215. [DOI:10.3390/ijms20174215] [PMID] [PMCID]
- Nowshehri JA, Bhat ZA, Shah MY. Blessings in disguise: Bio-functional benefits of grape seed extracts. Food Research International. 2015; 77(3):333-48. [DOI:10.1016/j.foodres.2015.08.026]
- Moon SW, Shin YU, Cho H, Bae SH, Kim HK. Effect of grape seed proanthocyanidin extract on hard exudates in patients with non-proliferative diabetic retinopathy. Medicine. 2019; 98(21):e15515. [DOI:10.1097/MD.0000000000015515] [PMID] [PMCID]
- Cheah KY, GS Howarth, Bastian SEP. Grape seed extract dose-responsively decreases disease severity in a rat model of mucositis; concomitantly enhancing chemotherapeutic effectiveness in colon cancer cells. PLOS One. 2014; 9(1):e85184. [DOI:10.1371/journal.pone.0085184] [PMID] [PMCID]
- Unusan N. Proanthocyanidins in grape seeds: An updated review of their health benefits and potential uses in the food industry. Journal of Functional Foods. 2020. 67: 103861. [DOI:10.1016/j.jff.2020.103861]
- Hemmati AA, Nazari Z, Samei M. A comparative study of grape seed extract and vitamin E effects on silica-induced pulmonary fibrosis in rats. Pulmonary Pharmacology & Therapeutics. 2008. 21(4):668-74. [DOI:10.1016/j.pupt.2008.04.004] [PMID]
- Salmabadi Z, Mohseni Kouchesfahani H, Parivar K, Karimzadeh L. Effect of grape seed extract on lipid profile and expression of interleukin-6 in polycystic ovarian syndrome wistar rat model. International Journal of Fertility & Sterility. 2017; 11(3):176-83. [DOI: 10.22074/ijfs.2017.5007] [PMID] [PMCID]
Type of Study: Original |
Subject:
Histology
Received: 2020/09/30 | Accepted: 2020/11/8 | Published: 2020/07/11
Received: 2020/09/30 | Accepted: 2020/11/8 | Published: 2020/07/11
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |




