Sun, Jul 13, 2025
Volume 17, Issue 1 (Winter & Spring 2020)
ASJ 2020, 17(1): 21-32 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Azari Z, Kherullahi Z, Mohammadghasemi F, Aghajany Nasab M, Hoseini F, Gazor R. Effect of the Aqueous and Hydro-Alcoholic Extracts of Viola odorata L. on Biochemical and Histologic Liver Parameters in Diabetic Wistar Rats. ASJ 2020; 17 (1) :21-32
URL: http://anatomyjournal.ir/article-1-249-en.html
URL: http://anatomyjournal.ir/article-1-249-en.html
Zoleikha Azari1 

 , Zahra Kherullahi1
, Zahra Kherullahi1 

 , Fahimeh Mohammadghasemi1
, Fahimeh Mohammadghasemi1 

 , Monireh Aghajany Nasab1
, Monireh Aghajany Nasab1 

 , Farideh Hoseini1
, Farideh Hoseini1 

 , Rouhollah Gazor1
, Rouhollah Gazor1 




 , Zahra Kherullahi1
, Zahra Kherullahi1 

 , Fahimeh Mohammadghasemi1
, Fahimeh Mohammadghasemi1 

 , Monireh Aghajany Nasab1
, Monireh Aghajany Nasab1 

 , Farideh Hoseini1
, Farideh Hoseini1 

 , Rouhollah Gazor1
, Rouhollah Gazor1 


1- Cellular and Molecular Research Center, Faculty of Medicine, Guilan University of Medical Sciences, Rasht, Iran.
Full-Text [PDF 847 kb]
(1401 Downloads)
| Abstract (HTML) (3824 Views)
Full-Text: (2279 Views)
1. Introduction
The prevalence of diabetes, as the most important endocrine disease, is increasing [1]. According to the World Health Organization (WHO), diabetes will be the seventh cause of death in 2030 [2]. In Iran, the prevalence of diabetes is on the rise, and according to forecasts, in 2025, 5.2 million Iranian people will be suffering from diabetes [3].
The initial stage of type 2 diabetes is characterized by insulin resistance in target tissues, especially the liver, skeletal muscle, and adipokines. Insulin resistance in these tissues results in the production of excess glucose by the liver [4]. This disease is associated with a wide range of liver diseases, such as an abnormal increase in liver enzymes, non-alcoholic fatty hepatic disease, liver cirrhosis, carcinoma of liver cells, and chronic liver disorder [5]. In diabetes, the balance between antioxidants and free radicals is distorted; thus, the process of emergence of diabetic side-effects accelerates, due to increased antioxidants in the body [6].
Oxidative stress results from an imbalance between the production of oxygen-free radicals and the defense of antioxidants in the body [7]. Chronic hyperglycemia can cause abundant and irreparable lesions in the eyes, nerves, kidneys, liver, as well as heart and vessels [8]. Treatments based on medicinal plants is typically cost-effective, more feasible, and more accessible, compared to chemical drugs-based treatments. Furthermore, in some cases, it has fewer adverse effects. Therefore, in diabetes treatment, it is necessary to find effective compounds with fewer side-effects. More than 1200 medicinal plants have been identified as effective in diabetes treatment [9].
Violets (Violaceae), which have a wide variety and wide application in Iran, have been previously studied. Liver protecting flavonoids in Viola odorata species and anti-diabetic and antioxidant properties of Viola Mandshurica species are documented [10, 11]. The prevalence of diabetes is surprisingly increasing around the world and its associated complications are serious public health issues. Therefore, the present study aimed to evaluate the effect of violets on the liver enzymes and liver histologic parameters in diabetic adult Wistar rats.
2. Materials and Methods
Violets (Viola odorata L.), with the age of two weeks, were collected from the northern regions of Iran (Javaherdeh, Mazandaran Province). Then, they were vacuum dried at 55°C (with final moisture of about 8%) and powdered. Hydro-alcoholic extract (ethanol 70%) and aqueous extract of it were prepared by the distillation method with the balloons and water vapor based on the protocol proposed by M. Rabbani et al. (with some changes) [12].
This research was conducted according to the ethical considerations by the National Institutes of Health (NIH) as well as the ethical and caring guidelines for laboratory animals (Code: IR. GUMS. REC. 1930349905).
In total, 64 adult Wistar rats (age: 6-8 weeks; weight: 220-250 gr) (obtained from Razi Institute, Iran) were maintained in plexiglas with mesh doors at 23±2°C, the relative humidity of 40%-60%, and 12:12-h light–dark cycle. In the first week, the rats had free access to water and food (using ad libitum protocol) to get accustomed to the lab environment.
To induce diabetes, streptozotocin (made in the USA, ZanosarTM) was injected Intraperitoneally (IP) at a dose of 45 mg/kg. Moreover, 72 hours after the injection, the rats’ blood samples were prepared through their tail tip. Then, their glucose levels were measured by glucometer, and rats with plasmatic glucose levels ˃250 mg/dL were considered as diabetic [13]. The injection was repeated only once again if they were not diabetic. Additionally, the day when injecting aqueous and hydro-alcoholic extracts was conducted, the body weight and blood glucose levels of rats were recorded; accordingly, those with <250 mg/dL were excluded from the study. The treatment of diabetic rats was performed IP by aqueous and hydro-alcoholic extracts of violets with the concentrations of 100, 200, and 400 mg/kg, for 4 weeks [10, 14, 15].
In total, 64 adult Wistar rats (56 diabetic and 8 healthy rats) were studied in 8 groups of 8 rats, as follows: group A: the healthy control group; group B: diabetic rats (prescribed Streptozotocin: 45 mg/kg); groups C-E: diabetic rats treated with the concentrations of 100, 200, and 400 mg/kg of aqueous extract, and group F-H: treated with the concentrations of 100, 200, and 400 mg/kg of hydro-alcoholic extract.
Blood sampling was performed for biochemical studies after 4 weeks of intervention initiation. In addition, after the anesthesia induction with ketamine-xylene, the liver tissue was removed, dissected for histological investigation, and stained by Hematoxylin and Eosin (H & E) and Periodic acid–Schiff (PAS) methods. In the H&E staining, the cells’ nucleus is purple, and the cytoplasm is pink. This staining was performed for histological purposes, including congestion, inflammatory cells, cytoplasm hyper eosinophilia, the accumulation of active Kupffer cells, and central venous changes. Moreover, the PAS staining was used to investigate glycogen reserves in hepatocytes. From each tissue, 5 slides, and in each slide, 10 fields with a 400× magnification were investigated.
Serum glucose level was measured by the serum glucose kit (obtained from MerckTM, Germany), according to the glucose oxidase method using the photometric method and on 546 nm wavelength.
Using the Bromocresol Green kit method (obtained from MerckTM, Germany), by a special measurement kit to the measurement of serum albumin, rats’ serum albumin was measured. Then, wavelength was investigated using the photometric method on 546 nm.
Serum of liver enzymes was measured by a special measurement kit (obtained from MerckTM, Germany) by the photometric method. The sensitivity of the measurement method for ALT, AST, and ALP was 4, 2, and 3 international units per liter, respectively.
The collected data were analyzed using the Kolmogorov-Smirnov (K-S) method to ensure the normality of data distribution. The obtained results were reported as mean and standard deviation. Comparing the results of 8 groups was performed using one-way Analysis of Variance (ANOVA). The inter-group comparison was conducted using the Tukey method. All statistical analyses were conducted by SPSS. P<0.05 were considered as significant.
3. Results
During injecting aqueous and hydro-alcoholic extracts, rats’ glucose and weight were periodically measured. All rats’ weight significantly decreased during the experiment, compared to the study onset (P<0.05). The rats’ weight and Fasting Blood Sugar (FBS) level, which were measured by blood samples from their tail tip and examined by the glucometer are reported in Table 1.
The prevalence of diabetes, as the most important endocrine disease, is increasing [1]. According to the World Health Organization (WHO), diabetes will be the seventh cause of death in 2030 [2]. In Iran, the prevalence of diabetes is on the rise, and according to forecasts, in 2025, 5.2 million Iranian people will be suffering from diabetes [3].
The initial stage of type 2 diabetes is characterized by insulin resistance in target tissues, especially the liver, skeletal muscle, and adipokines. Insulin resistance in these tissues results in the production of excess glucose by the liver [4]. This disease is associated with a wide range of liver diseases, such as an abnormal increase in liver enzymes, non-alcoholic fatty hepatic disease, liver cirrhosis, carcinoma of liver cells, and chronic liver disorder [5]. In diabetes, the balance between antioxidants and free radicals is distorted; thus, the process of emergence of diabetic side-effects accelerates, due to increased antioxidants in the body [6].
Oxidative stress results from an imbalance between the production of oxygen-free radicals and the defense of antioxidants in the body [7]. Chronic hyperglycemia can cause abundant and irreparable lesions in the eyes, nerves, kidneys, liver, as well as heart and vessels [8]. Treatments based on medicinal plants is typically cost-effective, more feasible, and more accessible, compared to chemical drugs-based treatments. Furthermore, in some cases, it has fewer adverse effects. Therefore, in diabetes treatment, it is necessary to find effective compounds with fewer side-effects. More than 1200 medicinal plants have been identified as effective in diabetes treatment [9].
Violets (Violaceae), which have a wide variety and wide application in Iran, have been previously studied. Liver protecting flavonoids in Viola odorata species and anti-diabetic and antioxidant properties of Viola Mandshurica species are documented [10, 11]. The prevalence of diabetes is surprisingly increasing around the world and its associated complications are serious public health issues. Therefore, the present study aimed to evaluate the effect of violets on the liver enzymes and liver histologic parameters in diabetic adult Wistar rats.
2. Materials and Methods
Violets (Viola odorata L.), with the age of two weeks, were collected from the northern regions of Iran (Javaherdeh, Mazandaran Province). Then, they were vacuum dried at 55°C (with final moisture of about 8%) and powdered. Hydro-alcoholic extract (ethanol 70%) and aqueous extract of it were prepared by the distillation method with the balloons and water vapor based on the protocol proposed by M. Rabbani et al. (with some changes) [12].
This research was conducted according to the ethical considerations by the National Institutes of Health (NIH) as well as the ethical and caring guidelines for laboratory animals (Code: IR. GUMS. REC. 1930349905).
In total, 64 adult Wistar rats (age: 6-8 weeks; weight: 220-250 gr) (obtained from Razi Institute, Iran) were maintained in plexiglas with mesh doors at 23±2°C, the relative humidity of 40%-60%, and 12:12-h light–dark cycle. In the first week, the rats had free access to water and food (using ad libitum protocol) to get accustomed to the lab environment.
To induce diabetes, streptozotocin (made in the USA, ZanosarTM) was injected Intraperitoneally (IP) at a dose of 45 mg/kg. Moreover, 72 hours after the injection, the rats’ blood samples were prepared through their tail tip. Then, their glucose levels were measured by glucometer, and rats with plasmatic glucose levels ˃250 mg/dL were considered as diabetic [13]. The injection was repeated only once again if they were not diabetic. Additionally, the day when injecting aqueous and hydro-alcoholic extracts was conducted, the body weight and blood glucose levels of rats were recorded; accordingly, those with <250 mg/dL were excluded from the study. The treatment of diabetic rats was performed IP by aqueous and hydro-alcoholic extracts of violets with the concentrations of 100, 200, and 400 mg/kg, for 4 weeks [10, 14, 15].
In total, 64 adult Wistar rats (56 diabetic and 8 healthy rats) were studied in 8 groups of 8 rats, as follows: group A: the healthy control group; group B: diabetic rats (prescribed Streptozotocin: 45 mg/kg); groups C-E: diabetic rats treated with the concentrations of 100, 200, and 400 mg/kg of aqueous extract, and group F-H: treated with the concentrations of 100, 200, and 400 mg/kg of hydro-alcoholic extract.
Blood sampling was performed for biochemical studies after 4 weeks of intervention initiation. In addition, after the anesthesia induction with ketamine-xylene, the liver tissue was removed, dissected for histological investigation, and stained by Hematoxylin and Eosin (H & E) and Periodic acid–Schiff (PAS) methods. In the H&E staining, the cells’ nucleus is purple, and the cytoplasm is pink. This staining was performed for histological purposes, including congestion, inflammatory cells, cytoplasm hyper eosinophilia, the accumulation of active Kupffer cells, and central venous changes. Moreover, the PAS staining was used to investigate glycogen reserves in hepatocytes. From each tissue, 5 slides, and in each slide, 10 fields with a 400× magnification were investigated.
Serum glucose level was measured by the serum glucose kit (obtained from MerckTM, Germany), according to the glucose oxidase method using the photometric method and on 546 nm wavelength.
Using the Bromocresol Green kit method (obtained from MerckTM, Germany), by a special measurement kit to the measurement of serum albumin, rats’ serum albumin was measured. Then, wavelength was investigated using the photometric method on 546 nm.
Serum of liver enzymes was measured by a special measurement kit (obtained from MerckTM, Germany) by the photometric method. The sensitivity of the measurement method for ALT, AST, and ALP was 4, 2, and 3 international units per liter, respectively.
The collected data were analyzed using the Kolmogorov-Smirnov (K-S) method to ensure the normality of data distribution. The obtained results were reported as mean and standard deviation. Comparing the results of 8 groups was performed using one-way Analysis of Variance (ANOVA). The inter-group comparison was conducted using the Tukey method. All statistical analyses were conducted by SPSS. P<0.05 were considered as significant.
3. Results
During injecting aqueous and hydro-alcoholic extracts, rats’ glucose and weight were periodically measured. All rats’ weight significantly decreased during the experiment, compared to the study onset (P<0.05). The rats’ weight and Fasting Blood Sugar (FBS) level, which were measured by blood samples from their tail tip and examined by the glucometer are reported in Table 1.
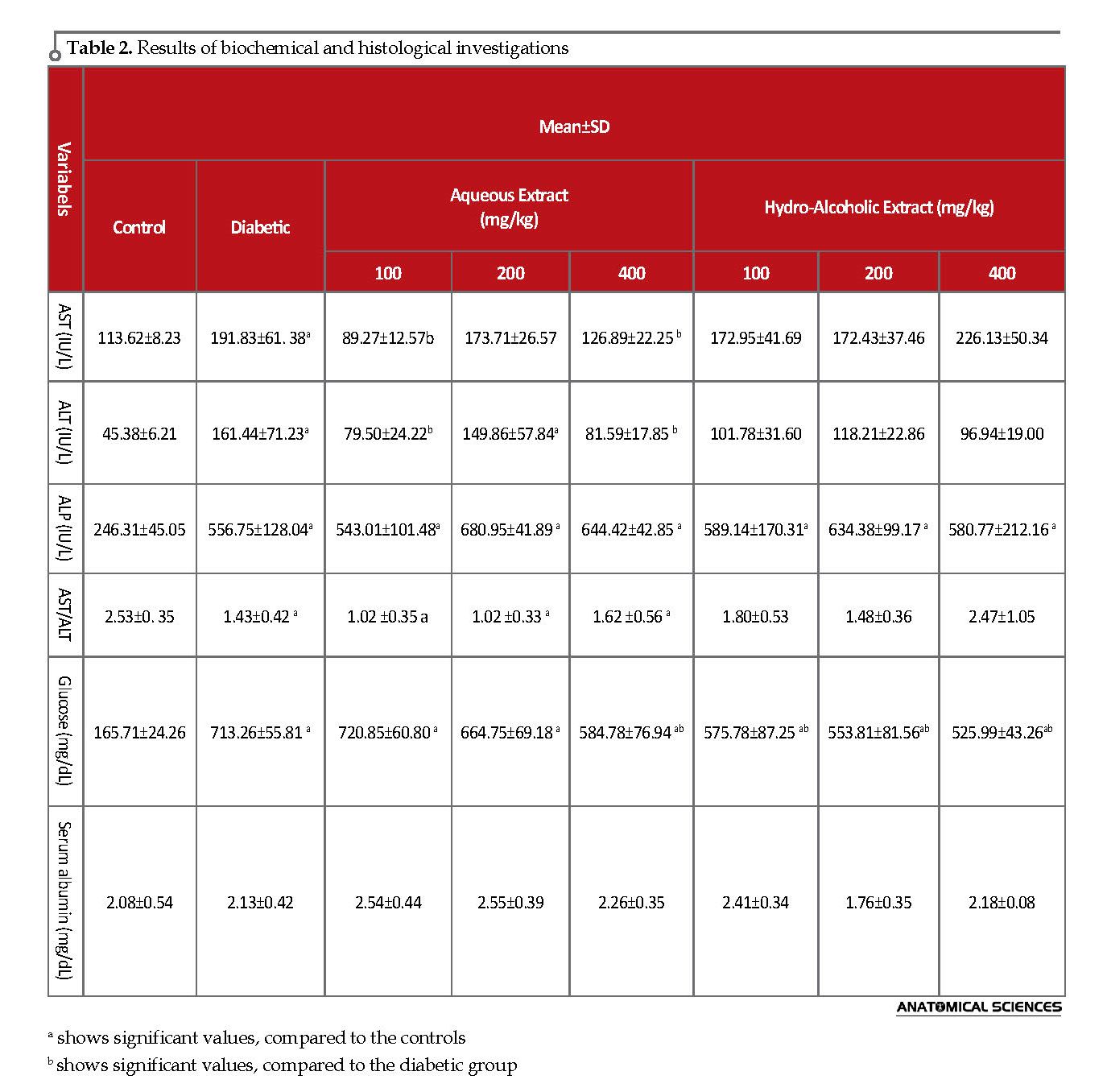
The obtained results indicated that AST significantly decreased in the concentrations of 100 and 400 mg/kg in the aqueous extract. In addition, ALT revealed a significant decrease in the concentration of 100 mg/kg in the aqueous and hydro-alcoholic extracts as well as the concentration of 400 mg/kg in the aqueous extract. Meanwhile, IP injection of different aqueous and hydro-alcoholic concentrations suggested no change in serum ALP. The AST/ALT ratio also significantly reduced by the IP injection of different concentrations of aqueous extract, compared to the control group (Table 2).
The present study revealed a significant decrease in the glucose level of the streptozotocin-diabetic group, compared with the controls. This finding confirmed the rats becoming diabetic (P<0.05). In addition, IP injection of 100, 200, and 400 mg/kg of hydro-alcoholic extract of violets, and the concentration of 400 mg/kg of the aqueous extract significantly decreased the serum glucose level in diabetic rats (P<0.001). Moreover, the albumin level of the streptozotocin-diabetic group remained unchanged, compared to the controls (Table 2).
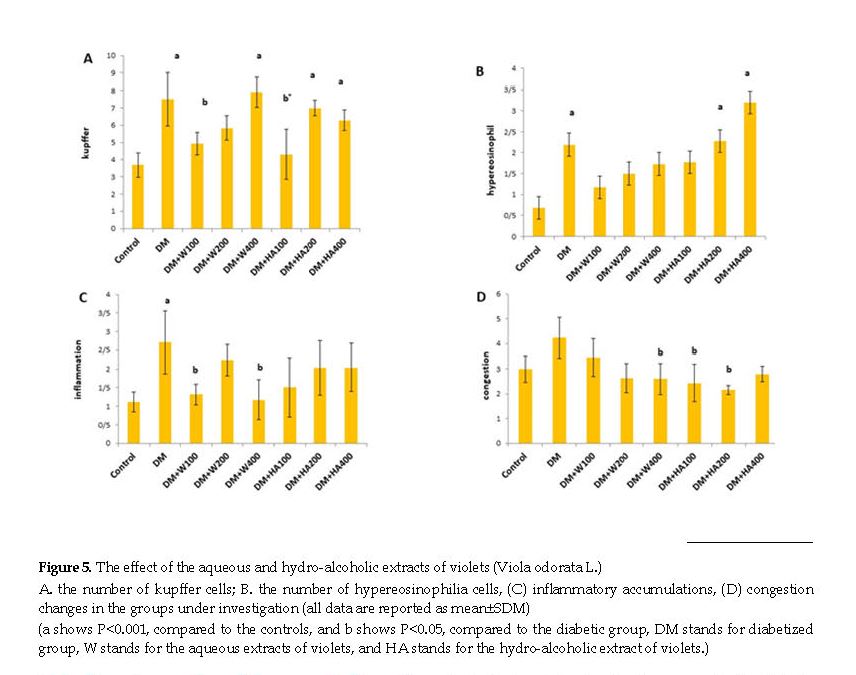
We observed the slides produced by the optical microscope in the control group, diabetic group, and diabetic group receiving different concentrations of the aqueous and hydro-alcoholic extracts of violet. The relevant data suggested a significant increase in the number of Kupffer cells in the diabetic group, compared to the controls. In addition, the IP injection of 100 mg/kg of the aqueous and hydro-alcoholic extracts of violet significantly decreased the number of Kupffer cells. However, the concentrations of 200 and 400 mg/kg of the aqueous and hydro-alcoholic extracts failed to change the number of Kupffer cells (Figure 1).
Reduction of rats’ hypereosinophilia hepatocyte cells at the concentration of 100 mg/kg
Reduction of rats’ hypereosinophilia hepatocyte cells at the concentration of 100 mg/kg
Observing the slides produced by the optical microscope demonstrated that the number of hypereosinophilia cells increased in the diabetic group, compared to the controls. In addition, the IP injection of 100 mg/kg of the aqueous extract of violet decreased the number of hypereosinophilia cells, compared to the diabetic group.
Reduction of inflammatory accumulations at the concentrations of 100 and 400 mg/kg of the aqueous extract
Observing the slides produced by the optical microscope suggested that the inflammation rate increased in the diabetic group, compared to the controls. The IP injection of 100 and 400 mg/kg of the aqueous extract of violet significantly reduced inflammatory accumulations, in comparison with the diabetic groups.
Reduction of congestion at the concentrations of 400 mg/kg of the aqueous extract and 100 and 200 mg/kg of the hydro-alcoholic extract
Observing the slides prepared by the optical microscope revealed that the congestion rate remained unchanged in the diabetic group, in comparison with the controls. The IP injection of 400 mg/kg of the aqueous extract and 100 and 200 of the hydro-alcoholic extract significantly reduced congestion.
Observing the slides prepared by the optical microscope suggested that the diabetic group remained the same, in comparison with the controls in the perimeter and the area of the central vein. However, the IP injection of 200 mg/kg of the aqueous extract and 100 mg/kg of the hydro-alcoholic extract of violet significantly decreased (P<0.05).
Observing the slides produced by the optical microscope demonstrated that the diabetic group remained unchanged, compared to the controls in glycogen deposits. Although the level of glycogen was increased by injecting the hydro-alcoholic extract, compared to the diabetic group, the changes were not significant.
4. Discussion
Researchers keep investigating the potential effects of plants in the treatment of diseases. In this study, histological changes and functional tests of the liver were evaluated in the diabetic rats treated with different concentrations of the aqueous and hydro-alcoholic extracts of violet.
The prescription of streptozotocin with 45 mg/kg concentration in 72 hours induced diabetes in male rats. Streptozotocin, after being absorbed by the pancreas beta cells, by causing changes in the DNA and production of the oxygen-reactive ROS, leads to the degradation of these cells, dysfunction of insulin production, increased blood glucose (hyperglycemia), and the suffering of type 1 diabetes [16]. The changes obtained from streptozotocin on the liver include the dilation of the arteries, irregularity of hepatocyte plates, hepatic fibrosis, and the reduction of glycogen deposits [17].
Reduction of inflammatory accumulations at the concentrations of 100 and 400 mg/kg of the aqueous extract
Observing the slides produced by the optical microscope suggested that the inflammation rate increased in the diabetic group, compared to the controls. The IP injection of 100 and 400 mg/kg of the aqueous extract of violet significantly reduced inflammatory accumulations, in comparison with the diabetic groups.
Reduction of congestion at the concentrations of 400 mg/kg of the aqueous extract and 100 and 200 mg/kg of the hydro-alcoholic extract
Observing the slides prepared by the optical microscope revealed that the congestion rate remained unchanged in the diabetic group, in comparison with the controls. The IP injection of 400 mg/kg of the aqueous extract and 100 and 200 of the hydro-alcoholic extract significantly reduced congestion.
Observing the slides prepared by the optical microscope suggested that the diabetic group remained the same, in comparison with the controls in the perimeter and the area of the central vein. However, the IP injection of 200 mg/kg of the aqueous extract and 100 mg/kg of the hydro-alcoholic extract of violet significantly decreased (P<0.05).
Observing the slides produced by the optical microscope demonstrated that the diabetic group remained unchanged, compared to the controls in glycogen deposits. Although the level of glycogen was increased by injecting the hydro-alcoholic extract, compared to the diabetic group, the changes were not significant.
4. Discussion
Researchers keep investigating the potential effects of plants in the treatment of diseases. In this study, histological changes and functional tests of the liver were evaluated in the diabetic rats treated with different concentrations of the aqueous and hydro-alcoholic extracts of violet.
The prescription of streptozotocin with 45 mg/kg concentration in 72 hours induced diabetes in male rats. Streptozotocin, after being absorbed by the pancreas beta cells, by causing changes in the DNA and production of the oxygen-reactive ROS, leads to the degradation of these cells, dysfunction of insulin production, increased blood glucose (hyperglycemia), and the suffering of type 1 diabetes [16]. The changes obtained from streptozotocin on the liver include the dilation of the arteries, irregularity of hepatocyte plates, hepatic fibrosis, and the reduction of glycogen deposits [17].
The liver significantly affects maintaining healthy concentrations of glucose after eating; thus, it is involved in diabetes mellitus [18]. Diabetes leads to morphological and morphometric changes in hepatocytes, which complies with our findings in terms of histological assessments. Most studies on the liver have focused on biochemical and physiological changes, and the histological aspects and liver structure are less considered. In this respect, our results suggested an increase in the number of Kupffer cells, hypereosinophilia, and lymphomas inflammation in the diabetic group. Furthermore, different concentrations of the aqueous and hydro-alcoholic extracts of violet reduced the number of Kupffer cells, hypereosinophilia cells, inflammation, and congestion, and increased glycogen deposits.
According to our results, the concentrations of 100 mg/kg of the aqueous and hydro-alcoholic extracts of violet significantly decreased the number of Kupffer cells. Additionally, 100 and 400 mg/kg of the aqueous extract significantly decreased inflammatory accumulations. Therefore, the aqueous and hydro-alcoholic extracts reduced the complications of diabetes, such as increased number of Kupffer cells, inflammation, and congestion.
According to our results, the concentrations of 100 mg/kg of the aqueous and hydro-alcoholic extracts of violet significantly decreased the number of Kupffer cells. Additionally, 100 and 400 mg/kg of the aqueous extract significantly decreased inflammatory accumulations. Therefore, the aqueous and hydro-alcoholic extracts reduced the complications of diabetes, such as increased number of Kupffer cells, inflammation, and congestion.
The deposits of glycogen granules inside hepatocytes of the cytoplasm remained unchanged in all the treated groups by PAS staining [19]. The increased activity of hepatic enzymes of serum, which reflects liver damage due to the increase of the activity of ALP, AST, and ALT enzymes, results from their leakage from hepatic cytosol into the bloodstream [20]. Increasing the protein catabolism along with gluconeogenesis and urea formation in diabetic patients may increase these transaminases in the blood [21]. In our study, AST, ALT, and ALP levels of serum significantly increased in diabetic rats. However, after the treatment of diabetic rats with aqueous and hydro-alcoholic extracts, ALT, AST, AST to ALT ratio, and serum glucose decreased in some concentrations.
The obtained results indicated that the IP injection of different concentrations of 400 and 100 mg/kg of the aqueous extract of violet, in comparison with the diabetic group, significantly decreased AST and ALT concentrations in the diabetic rats. The hydro-alcoholic extract also reduced the ALT level in the treated groups, compared to the diabetic group; however, the change was not significant. With the injection of the violet extract, no significant decrease was observed in the ALP level of the diabetic rats. Although the ALP level increased, the increasing trend continued in liver degradation, and violets had no favorable impact on this enzyme. This might be due to the short treatment period. However, it could have favorable effects on this parameter in long-term use.
Research has suggested diabetes-induced oxidative stress among major factors causing tissue damage and biochemical disturbances in diabetic patients’ body cells [22]. Hyperglycemia by glucose oxidation, protein glycation, and poly-alcohol metabolism activation can accelerate the production and the oxygen-reactive ROS, increase the oxidation of fats, DNA, and proteins in different tissues [23]. Producing free radicals is a cause of change in liver enzymes activity in patients with diabetes [24]. Serum albumin is exclusively produced by the liver cells and has a long half-life (18 to 20 days). Moreover, approximately 4% of it is decomposed daily and, the degradation rate of albumin is low; thus, serum albumin levels are not an appropriate indicator for mild or acute hepatic dysfunction [24].
In our study, there were no significant differences in the serum albumin level between the diabetic group and the controls. One possible mechanism related to the beneficial effects of violet extract on the histological and biochemical changes of the liver can be caused by the alkaloids, flavonoids, and natural polyphenolic compounds in this plant. Vessal et al. concluded that the IP prescription of some flavonoids in the diabetic rats with streptozotocin significantly decreased the serum glucose levels; i.e. dependent on concentration. However, the same flavonoids do not affect blood glucose concentration in healthy animals [25]. A part of the beneficial effects and hypoglycemic flavonoids can probably increase the hexozakinaz and glucokinase of liver activity and protect and even increase the density of Langerhans-adrenergic cells due to their antioxidant properties. Lee et al. investigated the physiological activities of Viola Mandshurica to measure its antioxidant properties by the total phenolic content.
The obtained results indicated that the IP injection of different concentrations of 400 and 100 mg/kg of the aqueous extract of violet, in comparison with the diabetic group, significantly decreased AST and ALT concentrations in the diabetic rats. The hydro-alcoholic extract also reduced the ALT level in the treated groups, compared to the diabetic group; however, the change was not significant. With the injection of the violet extract, no significant decrease was observed in the ALP level of the diabetic rats. Although the ALP level increased, the increasing trend continued in liver degradation, and violets had no favorable impact on this enzyme. This might be due to the short treatment period. However, it could have favorable effects on this parameter in long-term use.
Research has suggested diabetes-induced oxidative stress among major factors causing tissue damage and biochemical disturbances in diabetic patients’ body cells [22]. Hyperglycemia by glucose oxidation, protein glycation, and poly-alcohol metabolism activation can accelerate the production and the oxygen-reactive ROS, increase the oxidation of fats, DNA, and proteins in different tissues [23]. Producing free radicals is a cause of change in liver enzymes activity in patients with diabetes [24]. Serum albumin is exclusively produced by the liver cells and has a long half-life (18 to 20 days). Moreover, approximately 4% of it is decomposed daily and, the degradation rate of albumin is low; thus, serum albumin levels are not an appropriate indicator for mild or acute hepatic dysfunction [24].
In our study, there were no significant differences in the serum albumin level between the diabetic group and the controls. One possible mechanism related to the beneficial effects of violet extract on the histological and biochemical changes of the liver can be caused by the alkaloids, flavonoids, and natural polyphenolic compounds in this plant. Vessal et al. concluded that the IP prescription of some flavonoids in the diabetic rats with streptozotocin significantly decreased the serum glucose levels; i.e. dependent on concentration. However, the same flavonoids do not affect blood glucose concentration in healthy animals [25]. A part of the beneficial effects and hypoglycemic flavonoids can probably increase the hexozakinaz and glucokinase of liver activity and protect and even increase the density of Langerhans-adrenergic cells due to their antioxidant properties. Lee et al. investigated the physiological activities of Viola Mandshurica to measure its antioxidant properties by the total phenolic content.
Moreover, its antidiabetic activity was estimated with the activities of the inhibitors on α-amylase and α-glucosidase. They concluded that methanol extract has the highest amount of phenol content. Acetone extract represents a more powerful radical scavenging activity. The acetone extracts inhibitory activity against α-amylase and α-glucosidase is higher than 100% at a concentration of 1000 μg/mL. The obtained results suggested that V. Mandshurica may have potential anti-oxidant and antidiabetic activities [26].
The violet extract contains active morphologic factors, such as alkaloid, glycoside, tannin, steroid, terpenoid, saponins, flavonoid, methyl salicylate, mucilage, and vitamin C [27-29]. Some of these compounds seem to somehow reduce the severity of self-safe reactions and the inflammation process leading to beta-cell destruction. Thus, preventing residual cells’ destruction may provide adequate opportunity for the proliferation of these cells and the reconstruction of Langerhans. In addition, about 30 cyclotides of the aerial and root sections of V. Odorata were identified [30]. Several reports have indicated that the anti-diabetic property of medicinal plants is due to the presence of saponins [4]. Compounds containing saponins are hayperglismic antioxidants and increase insulin secretion [31]. The blue compounds of the VO reveal the existence of anthocyanin [32]. Anthocyanins are antibacterial, antihistamine, anti-allergic, anti-malarial, fat- and cholesterol-reducing, and anti-diabetic agents [4].
Empirical evidence has revealed that some effects of ethanol lead to the induction of metabolic processes, resulting in an increased production of ROS [33]. A study reported that ethanol and atrazine have toxic effects on rats’ liver and kidney and cause severe destructive effects on catalase activity and superoxide dismutase [34]. According to these studies, due to the destructive effects of alcohol on the liver tissue, the hydro-alcoholic extracts, unlike the aqueous extracts were unable to have more effect on liver enzymes.
The obtained results suggested that the prescription of the aqueous extract of violet in 100 and 400 mg/kg doses can improve the liver tissue in terms of cell count, inflammation, and congestion. Moreover, they could significantly decrease AST and ALT enzymes. The 400 mg/kg dose of the aqueous extract and all used doses of the hydro-alcoholic extract significantly decreased serum glucose levels in the diabetic rats.
Ethical Considerations
Compliance with ethical guidelines
All ethical principles were considered in this article.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed in preparing this article.
Conflict of interest
The authors declared no conflict of interest.
References
Wan Y, Garner J, Wu N, Phillip L, Han Y, McDaniel K, et al. Role of stem cells during diabetic liver injury. Journal of Cellular and Molecular Medicine. 2016; 20(2):195-203. [DOI:10.1111/jcmm.12723] [PMID] [PMCID]
Mathers CD, Loncar D. Projections of global mortality and burden of disease from 2002 to 2030. PLoS Medicine. 2006; 3(11):e442. [DOI:10.1371/journal.pmed.0030442] [PMID] [PMCID]
King H, Aubert RE, Herman WH. Global burden of diabetes, 1995-2025: Prevalence, numerical estimates, and projections. Diabetes Care. 1998; 21(9):1414-31. [DOI:10.2337/diacare.21.9.1414] [PMID]
Elekofehinti OO. Saponins: Anti-diabetic principles from medicinal plants: A review. Pathophysiology. 2015; 22(2):95-103. [DOI:10.1016/j.pathophys.2015.02.001] [PMID]
Leeds JS, Forman EM, Morley S, Scott AR, Tesfaye S, Sanders DS. Abnormal liver function tests in patients with type 1 diabetes mellitus: Prevalence, clinical correlations and underlying pathologies. Diabetic Medicine. 2009; 26(12):1235-41. [DOI:10.1111/j.1464-5491.2009.02839.x] [PMID]
Atalay M, Laaksonen DE. Diabetes, oxidative stress and physical exercise. Journal of Sports Science and Medicine. 2002; 1(1):1-14. [PMID] [PMCID]
Penckofer S, Schwertz D, Florczak K. Oxidative stress and cardiovascular disease in type 2 diabetes: The role of antioxidants and pro-oxidants. The Journal of Cardiovascular Nursing. 2002; 16(2):68-85. [DOI:10.1097/00005082-200201000-00007] [PMID]
Mayfield J. Diagnosis and classification of diabetes mellitus: New criteria. American Family Physician. 1998; 58(6):1355-62. [PMID]
Karim A, Sohail MN, Munir S, Sattar S. Pharmacology and phytochemistry of Pakistani herbs and herbal drugs used for treatment of diabetes. International Journal of Pharmacology. 2011; 7(4):419-39. [DOI:10.3923/ijp.2011.419.439]
Imran Qadir M, Ali M, Saleem M, Hanif M. Hepatoprotective activity of aqueous methanolic extract of Viola odorata against paracetamol-induced liver injury in mice. Bangladesh Journal of Pharmacology. 2014; 9:198-202. [DOI:10.3329/bjp.v9i2.18049]
Jo JH, Moon SH, Huh MK. Risk-benefit assessment between inhibition of angiotensin converting enzyme (ACE) and melanogenesis of medicinal herb. Viola Mandshurica. 2017: 4(9),85-29.
Rabbani M, Sajjadi SE, Jalali A. Hydroalcohol extract and fractions of Stachys lavandulifolia vahl: Effects on spontaneous motor activity and elevated plus maze behaviour. Phytotherapy Research. 2005; 19(10):854-8. [DOI:10.1002/ptr.1701] [PMID]
Kesari AN, Gupta RK, Singh SK, Diwakar S, Watal G. Hypoglycemic and antihyperglycemic activity of Aegle marmelos seed extract in normal and diabetic rats. Journal of Ethnopharmacology. 2006; 107(3):374-9. [DOI:10.1016/j.jep.2006.03.042] [PMID]
Alipanah H, Bigdeli MR, Esmaeili MA, Akbari ME. [Comparing the effect of hydro-alcoholic extract of Viola odorata and melatonin on tumor Growth and NF-kB, TNFR1, and VCAM-1 expression rates in 4T1 breast cancer model: An in vivo study (Persian)]. Journal of Mazandaran University of Medical Sciences. 2017; 27(147):25-40.
Mittal P, Gupta V, Goswami M, Thakur N, Bansal P. Phytochemical and pharmacological potential of viola odorata. International Journal of Pharmacognosy. 2015; 2(5):215-20. [DOI:10.13040/IJPSR.0975-8232.IJP.2(5).215-20]
Wright JR, Abraham C, Dickson BC, Yang H, Morrison CM. Streptozotocin dose-response curve in tilapia, a glucose-responsive teleost fish. General and Comparative Endocrinology. 1999; 114(3):431-40. [DOI:10.1006/gcen.1999.7269] [PMID]
Das AV, Padayatti PS, Paulose CS. Effect of leaf extract of Aegle marmelose (L.) Correa ex Roxb. On histological and ultrastructural changes in tissues of streptozotocin induced diabetic rats. Indian Journal of Experimental Biology. 1996; 34(4):341-5. [PMID]
Tanaka K, Nanbara S, Tanaka T, Koide H, Hayashi T. Aminotransferase activity in the liver of diabetic mice. Diabetes Research and Clinical Practice. 1988; 5(1):71-5. [DOI:10.1016/S0168-8227(88)80081-0]
Mahmoud MF, Sakr SM. Hepatoprotective effect of bee propolis in rat model of streptozotocin-induced diabetic hepatotoxicity: Light and electron microscopic study. Life Science Journal. 2013; 10(4):2048-54.
Navarro MC, Montilla MP, Martín A, Jiménez J, Utrilla MP. Free radical scavenger and antihepatotoxic activity of Rosmarinus tomentosus. Planta Medica. 1993; 59(04):312-4. [DOI:10.1055/s-2006-959688]
Vozarova B, Stefan N, Lindsay RS, Saremi A, Pratley RE, Bogardus C, et al. High alanine aminotransferase is associated with decreased hepatic insulin sensitivity and predicts the development of type 2 diabetes. Diabetes. 2002; 51(6):1889-95. [DOI:10.2337/diabetes.51.6.1889] [PMID]
Niedowicz DM, Daleke DL. The role of oxidative stress in diabetic complications. Cell Biochemistry and Biophysics. 2005; 43(2):289-330. [DOI:10.1385/CBB:43:2:289]
Osawa T, Kato Y. Protective role of antioxidative food factors in oxidative stress caused by hyperglycemia. Annals of the New York Academy of Sciences. 2005; 1043(1):440-51. [DOI:10.1196/annals.1333.050] [PMID]
Eze ED, Dawud FA, Zainab AA, Jimoh A, Malgwi IS, Isa AS. Preliminary studies of effects of vitamin C and zinc on some liver enzymes in alloxan-induced diabetic Wistar rats. Asian Journal of Medical Sciences. 2012; 4(1):17-22.
Vessal M, Hemmati M, Vasei M. Antidiabetic effects of quercetin in streptozocin-induced diabetic rats. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology. 2003; 135(3):357-64. [DOI:10.1016/S1532-0456(03)00140-6]
Lee BB, Park SR, Han CS, Han DY, Park EJ, Park HR, et al. Antioxidant activity and inhibition activity against α-amylase and α-glucosidase of Viola mandshurica extracts. Journal of the Korean Society of Food Science and Nutrition. 2008; 37(4):405-9. [DOI:10.3746/jkfn.2008.37.4.405]
Feyzabadi Z, Jafari F, Kamali SH, Ashayeri H, Badiee Aval Sh, Esfahani MM, et al. Efficacy of Viola odorata in treatment of chronic insomnia. Iranian Red Crescent Medical Journal. 2014; 16(12):e17511. [DOI:10.5812/ircmj.17511]
Stojković D, Glamočlija J, Ćirić A, Šiljegović J, Nikolić M, Soković M. Free radical scavenging activity of Viola odorata water extracts. Journal of Herbs, Spices & Medicinal Plants. 2011; 17(3):285-90. [DOI:10.1080/10496475.2011.603588]
Ireland DC, Colgrave ML, Craik DJ. A novel suite of cyclotides from Viola odorata: Sequence variation and the implications for structure, function and stability. Biochemical Journal. 2006; 400(1):1-12. [DOI:10.1042/BJ20060627] [PMID] [PMCID]
Giacco F, Brownlee M. Oxidative stress and diabetic complications. Circulation Research. 2010; 107(9):1058-70. [DOI:10.1161/CIRCRESAHA.110.223545] [PMID] [PMCID]
Karioti A, Furlan C, Vincieri FF, Bilia AR. Analysis of the constituents and quality control of Viola odorata aqueous preparations by HPLC-DAD and HPLC-ESI-MS. Analytical and Bioanalytical Chemistry. 2011; 399(4):1715-23. [DOI:10.1007/s00216-010-4473-2] [PMID]
Ghosh D, Konishi T. Anthocyanins and anthocyanin-rich extracts: Role in diabetes and eye function. Asia Pacific Journal of Clinical Nutrition. 2007; 16(S2):200-8. [PMID]
Molina MF, Sanchez-Reus I, Iglesias I, Benedi J. Quercetin, a flavonoid antioxidant, prevents and protects against ethanol-induced oxidative stress in mouse liver. Biological and Pharmaceutical Bulletin. 2003; 26(10):1398-402. [DOI:10.1248/bpb.26.1398] [PMID]
Abarikwu SO, Duru QC, Njoku RCC, Amadi BA, Tamunoibuomie A, Keboh E. Effects of co-exposure to atrazine and ethanol on the oxidative damage of kidney and liver in Wistar rats. Renal Failure. 2017; 39(1):588-96. [DOI:10.1080/0886022X.2017.1351373] [PMID] [PMCID]
The violet extract contains active morphologic factors, such as alkaloid, glycoside, tannin, steroid, terpenoid, saponins, flavonoid, methyl salicylate, mucilage, and vitamin C [27-29]. Some of these compounds seem to somehow reduce the severity of self-safe reactions and the inflammation process leading to beta-cell destruction. Thus, preventing residual cells’ destruction may provide adequate opportunity for the proliferation of these cells and the reconstruction of Langerhans. In addition, about 30 cyclotides of the aerial and root sections of V. Odorata were identified [30]. Several reports have indicated that the anti-diabetic property of medicinal plants is due to the presence of saponins [4]. Compounds containing saponins are hayperglismic antioxidants and increase insulin secretion [31]. The blue compounds of the VO reveal the existence of anthocyanin [32]. Anthocyanins are antibacterial, antihistamine, anti-allergic, anti-malarial, fat- and cholesterol-reducing, and anti-diabetic agents [4].
Empirical evidence has revealed that some effects of ethanol lead to the induction of metabolic processes, resulting in an increased production of ROS [33]. A study reported that ethanol and atrazine have toxic effects on rats’ liver and kidney and cause severe destructive effects on catalase activity and superoxide dismutase [34]. According to these studies, due to the destructive effects of alcohol on the liver tissue, the hydro-alcoholic extracts, unlike the aqueous extracts were unable to have more effect on liver enzymes.
The obtained results suggested that the prescription of the aqueous extract of violet in 100 and 400 mg/kg doses can improve the liver tissue in terms of cell count, inflammation, and congestion. Moreover, they could significantly decrease AST and ALT enzymes. The 400 mg/kg dose of the aqueous extract and all used doses of the hydro-alcoholic extract significantly decreased serum glucose levels in the diabetic rats.
Ethical Considerations
Compliance with ethical guidelines
All ethical principles were considered in this article.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed in preparing this article.
Conflict of interest
The authors declared no conflict of interest.
References
Wan Y, Garner J, Wu N, Phillip L, Han Y, McDaniel K, et al. Role of stem cells during diabetic liver injury. Journal of Cellular and Molecular Medicine. 2016; 20(2):195-203. [DOI:10.1111/jcmm.12723] [PMID] [PMCID]
Mathers CD, Loncar D. Projections of global mortality and burden of disease from 2002 to 2030. PLoS Medicine. 2006; 3(11):e442. [DOI:10.1371/journal.pmed.0030442] [PMID] [PMCID]
King H, Aubert RE, Herman WH. Global burden of diabetes, 1995-2025: Prevalence, numerical estimates, and projections. Diabetes Care. 1998; 21(9):1414-31. [DOI:10.2337/diacare.21.9.1414] [PMID]
Elekofehinti OO. Saponins: Anti-diabetic principles from medicinal plants: A review. Pathophysiology. 2015; 22(2):95-103. [DOI:10.1016/j.pathophys.2015.02.001] [PMID]
Leeds JS, Forman EM, Morley S, Scott AR, Tesfaye S, Sanders DS. Abnormal liver function tests in patients with type 1 diabetes mellitus: Prevalence, clinical correlations and underlying pathologies. Diabetic Medicine. 2009; 26(12):1235-41. [DOI:10.1111/j.1464-5491.2009.02839.x] [PMID]
Atalay M, Laaksonen DE. Diabetes, oxidative stress and physical exercise. Journal of Sports Science and Medicine. 2002; 1(1):1-14. [PMID] [PMCID]
Penckofer S, Schwertz D, Florczak K. Oxidative stress and cardiovascular disease in type 2 diabetes: The role of antioxidants and pro-oxidants. The Journal of Cardiovascular Nursing. 2002; 16(2):68-85. [DOI:10.1097/00005082-200201000-00007] [PMID]
Mayfield J. Diagnosis and classification of diabetes mellitus: New criteria. American Family Physician. 1998; 58(6):1355-62. [PMID]
Karim A, Sohail MN, Munir S, Sattar S. Pharmacology and phytochemistry of Pakistani herbs and herbal drugs used for treatment of diabetes. International Journal of Pharmacology. 2011; 7(4):419-39. [DOI:10.3923/ijp.2011.419.439]
Imran Qadir M, Ali M, Saleem M, Hanif M. Hepatoprotective activity of aqueous methanolic extract of Viola odorata against paracetamol-induced liver injury in mice. Bangladesh Journal of Pharmacology. 2014; 9:198-202. [DOI:10.3329/bjp.v9i2.18049]
Jo JH, Moon SH, Huh MK. Risk-benefit assessment between inhibition of angiotensin converting enzyme (ACE) and melanogenesis of medicinal herb. Viola Mandshurica. 2017: 4(9),85-29.
Rabbani M, Sajjadi SE, Jalali A. Hydroalcohol extract and fractions of Stachys lavandulifolia vahl: Effects on spontaneous motor activity and elevated plus maze behaviour. Phytotherapy Research. 2005; 19(10):854-8. [DOI:10.1002/ptr.1701] [PMID]
Kesari AN, Gupta RK, Singh SK, Diwakar S, Watal G. Hypoglycemic and antihyperglycemic activity of Aegle marmelos seed extract in normal and diabetic rats. Journal of Ethnopharmacology. 2006; 107(3):374-9. [DOI:10.1016/j.jep.2006.03.042] [PMID]
Alipanah H, Bigdeli MR, Esmaeili MA, Akbari ME. [Comparing the effect of hydro-alcoholic extract of Viola odorata and melatonin on tumor Growth and NF-kB, TNFR1, and VCAM-1 expression rates in 4T1 breast cancer model: An in vivo study (Persian)]. Journal of Mazandaran University of Medical Sciences. 2017; 27(147):25-40.
Mittal P, Gupta V, Goswami M, Thakur N, Bansal P. Phytochemical and pharmacological potential of viola odorata. International Journal of Pharmacognosy. 2015; 2(5):215-20. [DOI:10.13040/IJPSR.0975-8232.IJP.2(5).215-20]
Wright JR, Abraham C, Dickson BC, Yang H, Morrison CM. Streptozotocin dose-response curve in tilapia, a glucose-responsive teleost fish. General and Comparative Endocrinology. 1999; 114(3):431-40. [DOI:10.1006/gcen.1999.7269] [PMID]
Das AV, Padayatti PS, Paulose CS. Effect of leaf extract of Aegle marmelose (L.) Correa ex Roxb. On histological and ultrastructural changes in tissues of streptozotocin induced diabetic rats. Indian Journal of Experimental Biology. 1996; 34(4):341-5. [PMID]
Tanaka K, Nanbara S, Tanaka T, Koide H, Hayashi T. Aminotransferase activity in the liver of diabetic mice. Diabetes Research and Clinical Practice. 1988; 5(1):71-5. [DOI:10.1016/S0168-8227(88)80081-0]
Mahmoud MF, Sakr SM. Hepatoprotective effect of bee propolis in rat model of streptozotocin-induced diabetic hepatotoxicity: Light and electron microscopic study. Life Science Journal. 2013; 10(4):2048-54.
Navarro MC, Montilla MP, Martín A, Jiménez J, Utrilla MP. Free radical scavenger and antihepatotoxic activity of Rosmarinus tomentosus. Planta Medica. 1993; 59(04):312-4. [DOI:10.1055/s-2006-959688]
Vozarova B, Stefan N, Lindsay RS, Saremi A, Pratley RE, Bogardus C, et al. High alanine aminotransferase is associated with decreased hepatic insulin sensitivity and predicts the development of type 2 diabetes. Diabetes. 2002; 51(6):1889-95. [DOI:10.2337/diabetes.51.6.1889] [PMID]
Niedowicz DM, Daleke DL. The role of oxidative stress in diabetic complications. Cell Biochemistry and Biophysics. 2005; 43(2):289-330. [DOI:10.1385/CBB:43:2:289]
Osawa T, Kato Y. Protective role of antioxidative food factors in oxidative stress caused by hyperglycemia. Annals of the New York Academy of Sciences. 2005; 1043(1):440-51. [DOI:10.1196/annals.1333.050] [PMID]
Eze ED, Dawud FA, Zainab AA, Jimoh A, Malgwi IS, Isa AS. Preliminary studies of effects of vitamin C and zinc on some liver enzymes in alloxan-induced diabetic Wistar rats. Asian Journal of Medical Sciences. 2012; 4(1):17-22.
Vessal M, Hemmati M, Vasei M. Antidiabetic effects of quercetin in streptozocin-induced diabetic rats. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology. 2003; 135(3):357-64. [DOI:10.1016/S1532-0456(03)00140-6]
Lee BB, Park SR, Han CS, Han DY, Park EJ, Park HR, et al. Antioxidant activity and inhibition activity against α-amylase and α-glucosidase of Viola mandshurica extracts. Journal of the Korean Society of Food Science and Nutrition. 2008; 37(4):405-9. [DOI:10.3746/jkfn.2008.37.4.405]
Feyzabadi Z, Jafari F, Kamali SH, Ashayeri H, Badiee Aval Sh, Esfahani MM, et al. Efficacy of Viola odorata in treatment of chronic insomnia. Iranian Red Crescent Medical Journal. 2014; 16(12):e17511. [DOI:10.5812/ircmj.17511]
Stojković D, Glamočlija J, Ćirić A, Šiljegović J, Nikolić M, Soković M. Free radical scavenging activity of Viola odorata water extracts. Journal of Herbs, Spices & Medicinal Plants. 2011; 17(3):285-90. [DOI:10.1080/10496475.2011.603588]
Ireland DC, Colgrave ML, Craik DJ. A novel suite of cyclotides from Viola odorata: Sequence variation and the implications for structure, function and stability. Biochemical Journal. 2006; 400(1):1-12. [DOI:10.1042/BJ20060627] [PMID] [PMCID]
Giacco F, Brownlee M. Oxidative stress and diabetic complications. Circulation Research. 2010; 107(9):1058-70. [DOI:10.1161/CIRCRESAHA.110.223545] [PMID] [PMCID]
Karioti A, Furlan C, Vincieri FF, Bilia AR. Analysis of the constituents and quality control of Viola odorata aqueous preparations by HPLC-DAD and HPLC-ESI-MS. Analytical and Bioanalytical Chemistry. 2011; 399(4):1715-23. [DOI:10.1007/s00216-010-4473-2] [PMID]
Ghosh D, Konishi T. Anthocyanins and anthocyanin-rich extracts: Role in diabetes and eye function. Asia Pacific Journal of Clinical Nutrition. 2007; 16(S2):200-8. [PMID]
Molina MF, Sanchez-Reus I, Iglesias I, Benedi J. Quercetin, a flavonoid antioxidant, prevents and protects against ethanol-induced oxidative stress in mouse liver. Biological and Pharmaceutical Bulletin. 2003; 26(10):1398-402. [DOI:10.1248/bpb.26.1398] [PMID]
Abarikwu SO, Duru QC, Njoku RCC, Amadi BA, Tamunoibuomie A, Keboh E. Effects of co-exposure to atrazine and ethanol on the oxidative damage of kidney and liver in Wistar rats. Renal Failure. 2017; 39(1):588-96. [DOI:10.1080/0886022X.2017.1351373] [PMID] [PMCID]
Type of Study: Original |
Subject:
Histology
Received: 2019/03/21 | Accepted: 2019/11/23 | Published: 2020/01/1
Received: 2019/03/21 | Accepted: 2019/11/23 | Published: 2020/01/1
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |