Tue, Jul 15, 2025
Volume 17, Issue 1 (Winter & Spring 2020)
ASJ 2020, 17(1): 13-20 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Marzban Abbasabadi B, Hajian O, Rahmati S. Investigating the Morphometric Characteristics of Male and Female Zell Sheep Skulls for Sexual Dimorphism. ASJ 2020; 17 (1) :13-20
URL: http://anatomyjournal.ir/article-1-221-en.html
URL: http://anatomyjournal.ir/article-1-221-en.html
1- Department of Basic Sciences, Faculty of Veterinary Medicine, Amol University of Special Modern Technologies, Amol, Iran.
2- School of Veterinary Medicine, Babol Branch, Islamic Azad University, Babol, Iran.
2- School of Veterinary Medicine, Babol Branch, Islamic Azad University, Babol, Iran.
Full-Text [PDF 572 kb]
(1945 Downloads)
| Abstract (HTML) (4565 Views)
3. Results
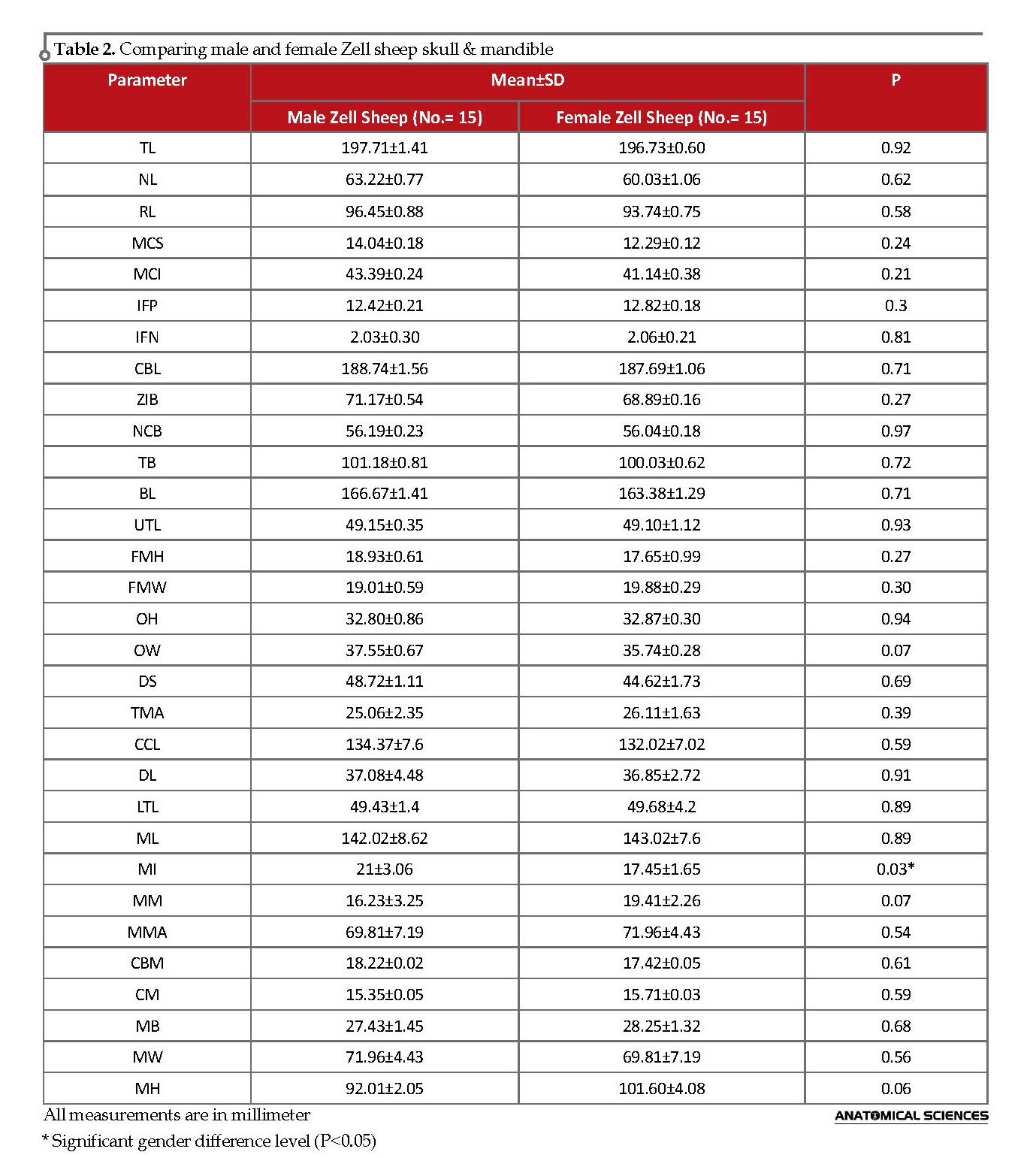
The descriptive analysis results (in mm) are presented in Table 2. The skull weight was significantly higher in males. However, no skull-related parameter indicated a significant difference between genders, except for the MI of the mandible; i.e. significantly higher in males, and its accuracy was equal to 83.33%.
4. Discussion
Dimorphism-based gender determination has been performed on most human bones. Reiches stated that applying some study methods occurs through two main approaches, as follows: by the report and description of the bones’ morphology, and by the values obtained using morphometry; or in other words, the measurements of these bones [4].
Abramov and Tumanov (2003) reported that zygomatic breadth and interorbital width were adequate for 96.5% correct classification of European mink Mustela lutreola. The male skull of M. lutreola is characterized by a relatively high neurocranium, widely arranged zygomatic arches, a wide rostrum, and with wider auditory bullae and higher mandibles [24].
Full-Text: (4080 Views)
1. Introduction
The skull is the major element of the skeleton, indicating taxonomic affiliation, and providing information on selection-induced changes in animals [1]. The craniometry is the foundation of clinical and surgical practices [2]. Similarly, different foramina of the skull are of clinical importance in regional anesthesia around the head [3]. The dimorphism of the skull and the pelvis has been highlighted by many authors. Krogman and Íscan stated that gender and race determination in a collection of 750 human skeletons with the presence of pelvis bone or skull (reliability: 95% and 92%, respectively) are possible. They have also reported that using the skull and pelvis bones could increase the examination reliability to 98%. This demonstrates the importance of these regions in gender determination [4].
Numerous comparative morphological and morphometrical studies have been performed on the skull anatomy in many mammalians to detect the distinguishing features of these species [5-8]. Moreover, there are other studies about gender-related characteristics in various animals, including elephants (Asian, Elephas maximus, and African, Loxodonta africana), raccoon dog (Nyctereutes procyonoides), badger (Meles meles), fox (Vulpes vulpes), dog (Canis lupus familiaris), and feline (Felis catus) [9-15]. However, gender identification in the sheep via bone morphometry is limited. Moreover, there is only one publication available on sexual dimorphism in sheep [16].
The Zell breed is the only thin-tailed sheep in Iran with a 10-12 cm long tail. This small sheep has a great ability to walk in the mountains and foothills due to its low body weight (40-45 kg) and fairly tall limbs. Zell sheep greatly impacts sheep production in its home area, north of Iran. In addition, it is the only local breed; i.e. suitable for crossbreeding [17-19]. Therefore, the present study aimed to identify the craniometric traits and sexual dimorphism in these characteristics on the male and female Zell sheep.
2. Material and Methods
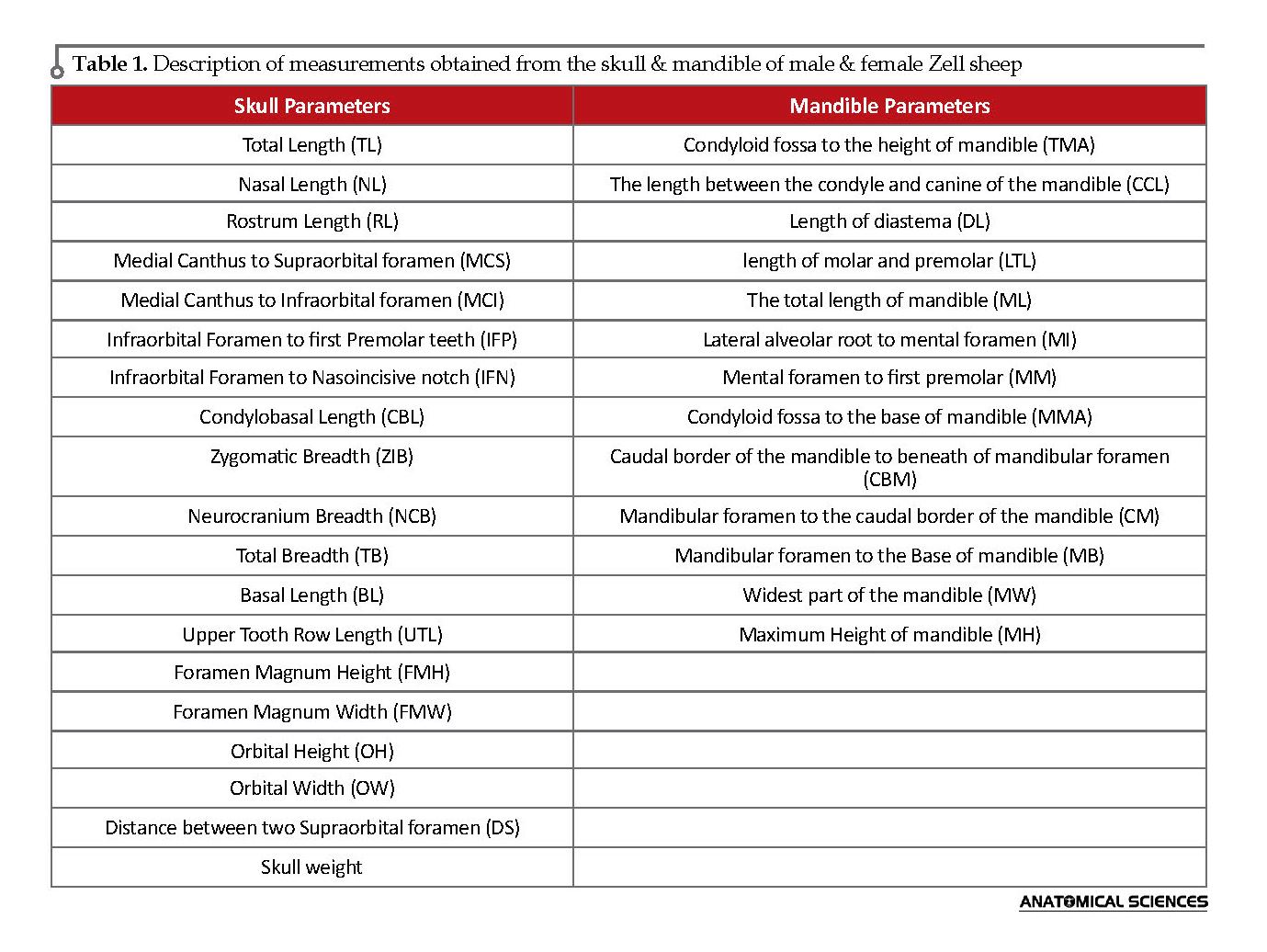
In total, 30 skulls (15 adult males and 15 adult females) of Zell sheep were selected from a local slaughterhouse. They were selected based on apparent good health conditions and the lack of skeletal deformities, pathologic lesions, and damages. The skulls were processed by hot water maceration, according to the standard previously reported techniques. Moreover, the mandible was dissociated from temporomandibular joint. All the specimens were coded and documented using a digital camera (Canon PowerShot SD790IS 10MP Digital Camera) [9].
The skull is the major element of the skeleton, indicating taxonomic affiliation, and providing information on selection-induced changes in animals [1]. The craniometry is the foundation of clinical and surgical practices [2]. Similarly, different foramina of the skull are of clinical importance in regional anesthesia around the head [3]. The dimorphism of the skull and the pelvis has been highlighted by many authors. Krogman and Íscan stated that gender and race determination in a collection of 750 human skeletons with the presence of pelvis bone or skull (reliability: 95% and 92%, respectively) are possible. They have also reported that using the skull and pelvis bones could increase the examination reliability to 98%. This demonstrates the importance of these regions in gender determination [4].
Numerous comparative morphological and morphometrical studies have been performed on the skull anatomy in many mammalians to detect the distinguishing features of these species [5-8]. Moreover, there are other studies about gender-related characteristics in various animals, including elephants (Asian, Elephas maximus, and African, Loxodonta africana), raccoon dog (Nyctereutes procyonoides), badger (Meles meles), fox (Vulpes vulpes), dog (Canis lupus familiaris), and feline (Felis catus) [9-15]. However, gender identification in the sheep via bone morphometry is limited. Moreover, there is only one publication available on sexual dimorphism in sheep [16].
The Zell breed is the only thin-tailed sheep in Iran with a 10-12 cm long tail. This small sheep has a great ability to walk in the mountains and foothills due to its low body weight (40-45 kg) and fairly tall limbs. Zell sheep greatly impacts sheep production in its home area, north of Iran. In addition, it is the only local breed; i.e. suitable for crossbreeding [17-19]. Therefore, the present study aimed to identify the craniometric traits and sexual dimorphism in these characteristics on the male and female Zell sheep.
2. Material and Methods
In total, 30 skulls (15 adult males and 15 adult females) of Zell sheep were selected from a local slaughterhouse. They were selected based on apparent good health conditions and the lack of skeletal deformities, pathologic lesions, and damages. The skulls were processed by hot water maceration, according to the standard previously reported techniques. Moreover, the mandible was dissociated from temporomandibular joint. All the specimens were coded and documented using a digital camera (Canon PowerShot SD790IS 10MP Digital Camera) [9].
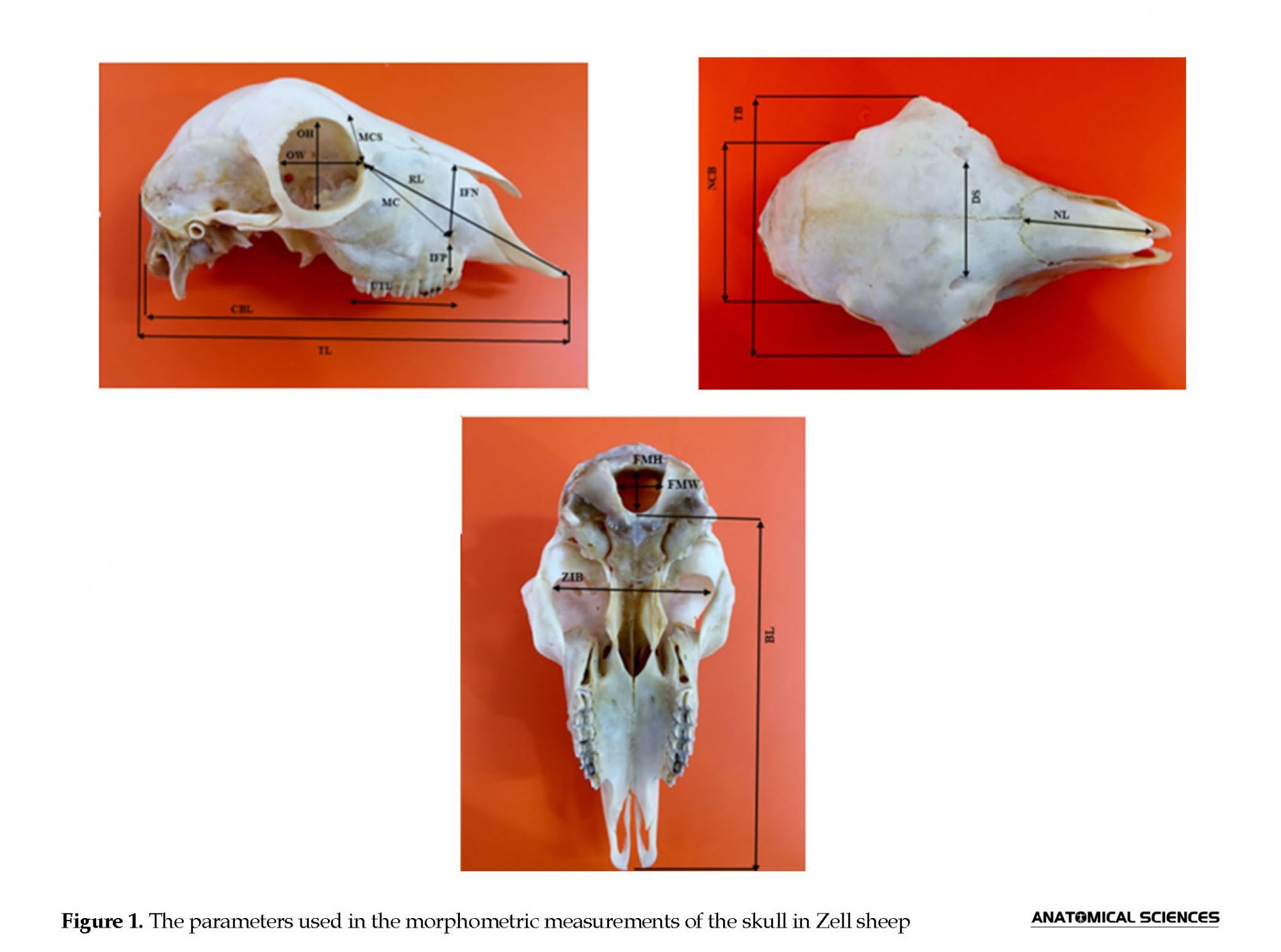
Subsequently, 19 parameters in the skull and 13 parameters in the mandible were calculated based on Onuk (2013) and Pitakarnnop et al. (2017), using digital ruler caliper (Digimatic Caliper, Japan) (Table 1) (Figures 1 & 2) [5, 14]. All measurements and observations were blinded, and each bone was evaluated twice for gender identification. The obtained data were analyzed in SPSS using the Independent Samples t-test. Additionally, the significance level was considered at P<0.05. The collected data are presented as Mean±SEM.
3. Results
The descriptive analysis results (in mm) are presented in Table 2. The skull weight was significantly higher in males. However, no skull-related parameter indicated a significant difference between genders, except for the MI of the mandible; i.e. significantly higher in males, and its accuracy was equal to 83.33%.
4. Discussion
Dimorphism-based gender determination has been performed on most human bones. Reiches stated that applying some study methods occurs through two main approaches, as follows: by the report and description of the bones’ morphology, and by the values obtained using morphometry; or in other words, the measurements of these bones [4].
Numerous studies signified the skull as one of the best skeleton parts to determine gender in humans and evaluated its gender differences [4]. In animals, despite many studies on skull gross anatomy, only a few has evaluated the gender-related differences [5, 6, 19-22]. Therefore, we investigated the morphometric characteristics of male and female Zell sheep. As per the obtained data, there was no significant difference in the parameters as mentioned earlier; however, the distance of lateral alveolar root to Mental Foramen (MI) was significantly higher in male Zell sheep (Table 2). Pares Casanova examined the magnitude of sexual dimorphism in a relict sheep from Catalunya (NE Spain), called Fardasca [16].Similar to our results, they suggested that the breed was not cranially dimorphic, and gender determination using landmarks such as skull shape and size was likely to yield poor results. In 1989, Jaslow studied the sexual dimorphism of cranial suture complexity in wild sheep. According to their results, most facial sutures were not sexually dimorphic; however, maxillojugal and jugolacrimal, had greater complexity in males, than in females. She suggested horn clashing as the most significant force that might be transmitted through the facial region of rams to develop this complexity. In females, the increased complexity of sutures during ontogeny was predicted by variables measuring the growth of the skull, brain, or face; while in males, changes in complexity were best predicted by variables representing mechanical loading and frontal bone growth [23].
Abramov and Tumanov (2003) reported that zygomatic breadth and interorbital width were adequate for 96.5% correct classification of European mink Mustela lutreola. The male skull of M. lutreola is characterized by a relatively high neurocranium, widely arranged zygomatic arches, a wide rostrum, and with wider auditory bullae and higher mandibles [24].
Farhadnia et al. (2014) explored the skull of Persian leopard (Panthera pardus saxicolor). They observed significant inter-sexual differences in the samples’ skull size. Their study also revealed that inter-sexual differentiation was also remarkable when comparing morphometric body measurements in adults. Their achieved data indicated that males have a larger head mass and longer body; however, there were no gender-specific differences in subadults [25].
Pitakarnnop et al. (2017) reported that no parameters from the skull demonstrated a significant difference between males and females cats. However, one parameter, Masseteric Moment Arm (MMA), was significantly higher in males with an accuracy of 64.9%. Similar to our study, a mandibular parameter was significantly different between males and females; although this parameter was MI (83.33% accuracy) in the present study [14].
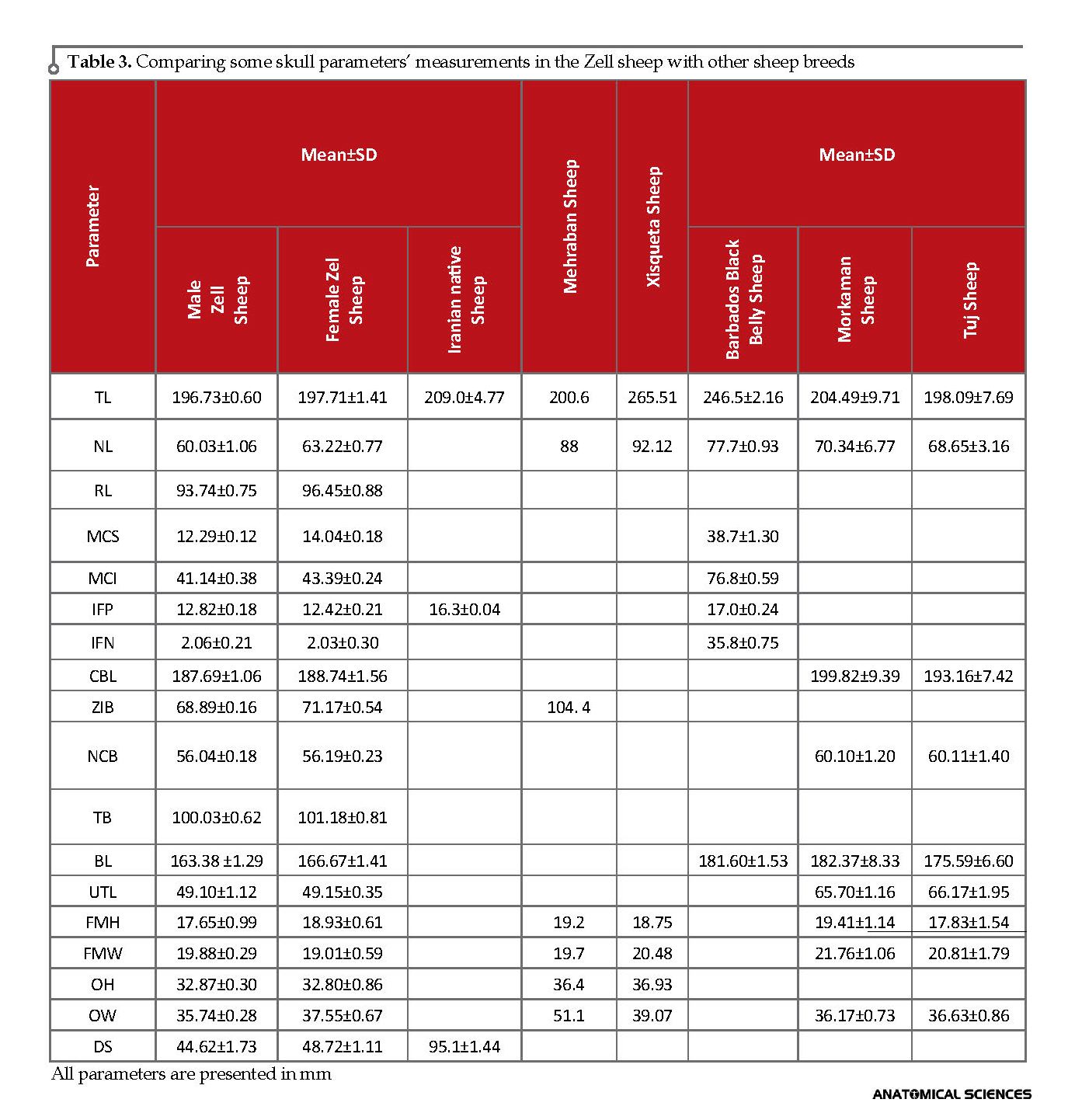
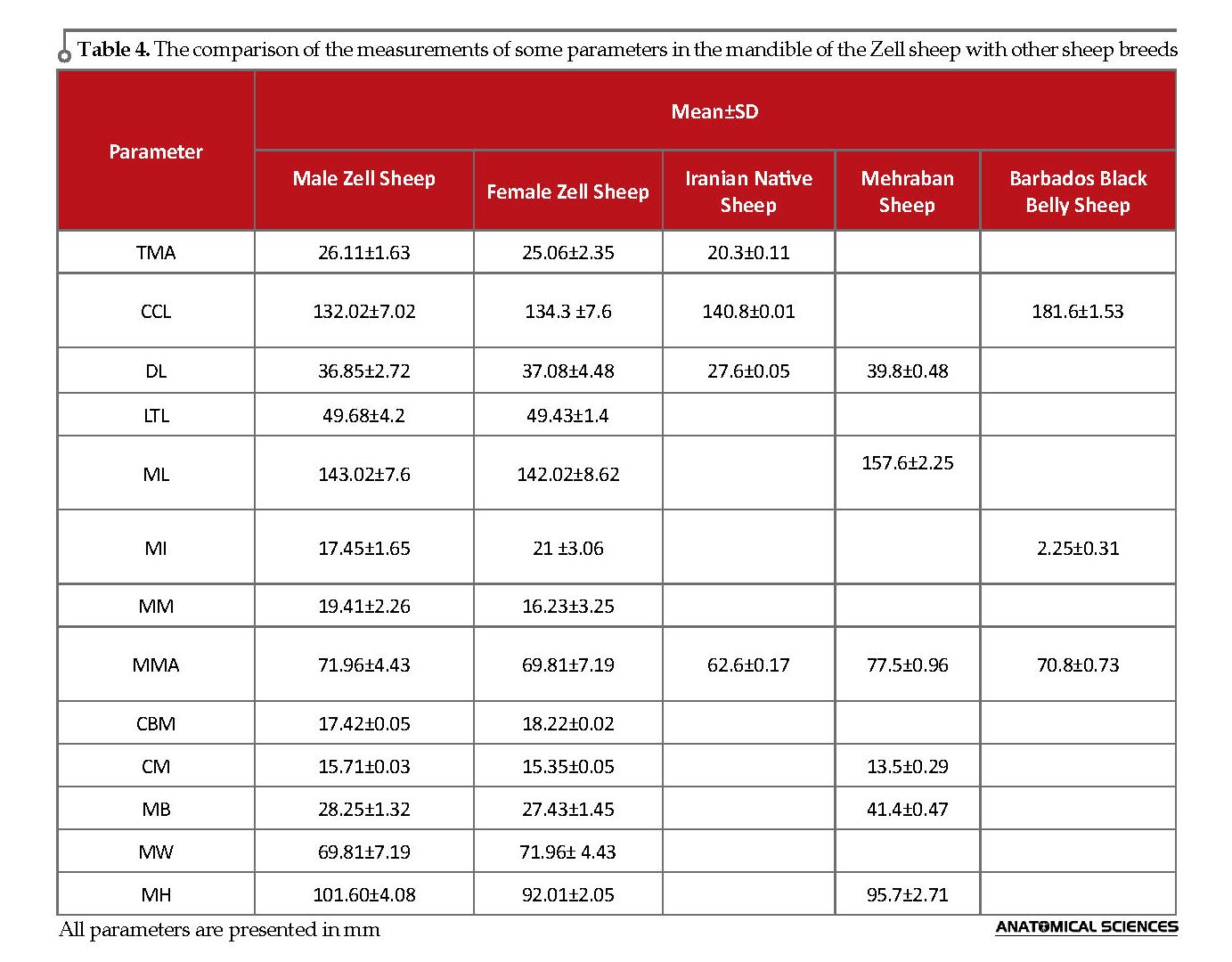
Moreover, many data such as TL, NL, MCS, MCI, CBL, ZIB, NCB, BL, UTL, FMH, OH, OW, DS, CCL, and Ml of Zell sheep was less, compared to the other studied sheep breeds, including Iranian breeds Therefore, we consider the Zell sheep breed as the smallest sheep in Iran and among the other studied sheep breeds (Table 3, 4) [3, 23, 24, 26-28].
The current study suggested that the distance of lateral alveolar root to mental foramen on mandible can be an appropriate landmark in gender estimation in Zell sheep. It also identified the skull of Zell sheep as the smallest studied sheep skull.
Ethical Considerations
Compliance with ethical guidelines
There was no ethical considerations to be considered in this research.
Funding
This research work has been supported by a research grant from the Amol University of Special Modern Technologies, Amol, Iran.
Authors' contributions
Conceptualization: Behrokh Marzban Abbasabadi; Methodology: Behrokh Marzban Abbasabadi; Investigation: All authors; Writing-review & editing: All authors; Funding Acquisition: Behrokh Marzban Abbasabadi; Supervision: Behrokh Marzban Abbasabadi.
Conflict of interest
The authors stated no conflicts of interest.
References
Brünner H, Lugon-Moulin N, Balloux F, Fumagalli L, Hausser J. A taxonomical re-evaluation of the Valais chromosome race of the common shrewSorex araneus (Insectivora: Soricidae). Acta Theriologica. 2002; 47(3):245-75. [DOI:10.1007/BF03194146]
Karimi I, Onar V, Pazvant G, Hadipour M, Mazaheri Y. The cranial morphometric and morphologic characteristics of Mehraban sheep in Western Iran. Global Veterinaria. 2011; 6(2):111-7.
Paiva LASD, Segre M. Sexing the human skull through the mastoid process. Revista do Hospital das Clínicas. 2003; 58(1):15-20. [DOI:10.1590/S0041-87812003000100004]
Onuk B, Kabak M, Atalar K. Anatomic and craniometric factors in differentiating roe deer (Capreolus capreolus) from sheep (Ovis aries) and goat (Capra hircus) skulls. Archives of Biological Sciences. 2013; 65(1):133-41. [DOI:10.2298/ABS1301141M]
Shawulu JC, Kwari HD, Olopade JO. Morphology of the bones of the skull in the Sahel ecotypes of goats (Capra hircus) in Nigeria. Journal of Animal and Veterinary Advances. 2011; 114(4):1-3. [PMID]
Alsafy MA, El-gendy SA, Abumandour MM. Computed tomography and gross anatomical studies on the head of one-humped camel (Camelus dromedarius). The Anatomical Record. 2014; 297(4):630-42. [DOI:10.1002/ar.22865] [PMID]
de la Barra R, Latorre E, Martínez ME, Calderón C. morphostructural differentiation and variability of merino sheep breed under sustained directional selection. International Journal of Morphology. 2014; 32(3):1069-73. [DOI:10.4067/S0717-95022014000300052]
Todd NE. Qualitative comparison of the cranio-dental osteology of the extant elephants, Elephas Maximus (Asian elephant) and Loxodonta Africana (African elephant). The Anatomical Record: Advances in Integrative Anatomy and Evolutionary Biology. 2010; 293(1):62-73. [DOI:10.1002/ar.21011] [PMID]
Nganvongpanit K, Siengdee P, Buddhachat K, Brown JL, Klinhom S, Pitakarnnop T, et al. Anatomy, histology and elemental profile of long bones and ribs of the Asian elephant (Elephas maximus). Anatomical Science International. 2017; 92(4):554-68. [DOI:10.1007/s12565-016-0361-y] [PMID]
Hidaka S, Matsumoto M, Hiji H, Ohsako S, Nishinakagava H. Morphology and morphometry of skulls of raccoon dogs, Nyctereutes procyonoides and badgers, Meles meles. Journal of Veterinary Medical Science. 1998; 60(2):161-7. [DOI:10.1292/jvms.60.161] [PMID]
Jurgelėnas E. Osteometric analysis of the pelvic bones and sacrum of the red fox and raccoon dog. Veterinarija ir Zootechnika. 2015; 70(92):42-7.
Kieser JA, Groeneveld HT. Mandibulodental allometry in the African wild dog, Lycaon pictus. Journal of Anatomy. 1992; 181(Pt 1):133-7. [PMID] [PMCID]
Pitakarnnop T, Buddhachat K, Euppayo T, Kriangwanich W, Nganvongpanit K. Feline (Felis catus) skull and pelvic morphology and morphometry: Gender-related difference? Anatomia, Histologia, Embryologia. 2017; 46(3):294-303. [DOI:10.1111/ahe.12269] [PMID]
Sicuro FL, Oliveira LF. Skull morphology and functionality of extant Felidae (Mammalia: Carnivora): A phylogenetic and evolutionary perspective. Zoological Journal of the Linnean Society. 2011; 161(2):414-62. [DOI:10.1111/j.1096-3642.2010.00636.x]
Pares-Casanova PM. Reduced skull sexual dimorphism in a local sheep breed. Iranian Journal of Applied Animal Science. 2014; 4(3):643-6.
Kashan NE, Azar GM, Afzalzadeh A, Salehi A. Growth performance and carcass quality of fattening lambs from fat-tailed and tailed sheep breeds. Small Ruminant Research. 2005; 60(3):267-71. [DOI:10.1016/j.smallrumres.2005.01.001]
Marzban Abbasabadi B, Kochakzadeh H, Kaveh Aski A. Evaluating gross anatomy of cervix in Zel sheep. Anatomical Sciences Journal. 2017; 14(3):115-20.
Sarma K. Morphological and craniometrical studies on the skull of Kagani goat (Capra hircus) of Jammu Region/Estudios morfologico y craniometrico de la cabra Kagani (Capra hircus) de la Region de Jammu. International Journal of Morphology. 2006; 24(3):449-56. [DOI:10.4067/S0717-95022006000400025]
Monfared AL, Naji H, Sheibani MT. Applied anatomy of the head region of the Iranian native goats (Capra hircus). Global Veternaria. 2013; 10(1):60-4. [DOI:10.5829/idosi.gv.2013.10.1.71133]
Saber AS, Caceci T, Gummow B, Johns K. Morphometric Studies on the Skull of the Australian Domestic Cat (F. catus) and its Clini-cal Implications for Regional Anesthesia. Journal of Veterinary Anatomy. 2016; 9(1):1-24.
Saber AS, Gummow B. Skull morphometry of the lion (Panthera leo), dog (Canis lupus familiaris) and cat (Felis catus). Journal of Veterinary Anatomy. 2015; 8(1):13-30.[DOI:10.21608/JVA.2015.44849]
JASLOW CR. Sexual dimorphism of cranial suture complexity in wild sheep (Ovis orientalis). Zoological Journal of the Linnean Society. 1989; 95(4):273-84. [DOI:10.1111/j.1096-3642.1989.tb02312.x]
Abramov AV, Tumanov IL. Sexual dimorphism in the skull of the European minkMustela lutreola from NW part of Russia. Acta Theriologica. 2003; 48(2):239-46. [DOI:10.1007/BF03194163]
Farhadinia MS, Kaboli M, Karami M, Farahmand H. Patterns of sexual dimorphism in the Persian Leopard (Panthera pardus saxicolor) and implications for sex differentiation. Zoology in the Middle East. 2014; 60(3):195-207. [DOI:10.1080/09397140.2014.939813]
Parés Casanova PM, Sarma K, Jordana i Vidal J. On biometrical aspects of the cephalic anatomy of Xisqueta sheep (Catalunya, Spain). International Journal of Morphology. 2010; 28(2):347-51. [DOI:10.4067/S0717-95022010000200001]
Özcan S, Aksoy G, Kürtül İ, Aslan K, Özüdoğru Z. A comparative morphometric study on the skull of the Tuj and Morkaraman sheep. Kafkas Üniversitesi Veteriner Fakültesi Dergisi. 2010; 16(1):111-4. [DOI:10.9775/kvfd.2009.518]
Karimi I, Hadipour M, Nikbakht P, Motamedi S. The lower jawbone of Mehreban sheep: A descriptive morphometric approach. World’s Veterinary Journal. 2011; 2:57-60.
Mohamed R, Driscoll M, Mootoo N. Clinical anatomy of the skull of the barbados black belly sheep in trinidad. International Journal of Research in Medical Sciences. 2016; 2(8):8-19.
Moreover, many data such as TL, NL, MCS, MCI, CBL, ZIB, NCB, BL, UTL, FMH, OH, OW, DS, CCL, and Ml of Zell sheep was less, compared to the other studied sheep breeds, including Iranian breeds Therefore, we consider the Zell sheep breed as the smallest sheep in Iran and among the other studied sheep breeds (Table 3, 4) [3, 23, 24, 26-28].
The current study suggested that the distance of lateral alveolar root to mental foramen on mandible can be an appropriate landmark in gender estimation in Zell sheep. It also identified the skull of Zell sheep as the smallest studied sheep skull.
Ethical Considerations
Compliance with ethical guidelines
There was no ethical considerations to be considered in this research.
Funding
This research work has been supported by a research grant from the Amol University of Special Modern Technologies, Amol, Iran.
Authors' contributions
Conceptualization: Behrokh Marzban Abbasabadi; Methodology: Behrokh Marzban Abbasabadi; Investigation: All authors; Writing-review & editing: All authors; Funding Acquisition: Behrokh Marzban Abbasabadi; Supervision: Behrokh Marzban Abbasabadi.
Conflict of interest
The authors stated no conflicts of interest.
References
Brünner H, Lugon-Moulin N, Balloux F, Fumagalli L, Hausser J. A taxonomical re-evaluation of the Valais chromosome race of the common shrewSorex araneus (Insectivora: Soricidae). Acta Theriologica. 2002; 47(3):245-75. [DOI:10.1007/BF03194146]
Karimi I, Onar V, Pazvant G, Hadipour M, Mazaheri Y. The cranial morphometric and morphologic characteristics of Mehraban sheep in Western Iran. Global Veterinaria. 2011; 6(2):111-7.
Paiva LASD, Segre M. Sexing the human skull through the mastoid process. Revista do Hospital das Clínicas. 2003; 58(1):15-20. [DOI:10.1590/S0041-87812003000100004]
Onuk B, Kabak M, Atalar K. Anatomic and craniometric factors in differentiating roe deer (Capreolus capreolus) from sheep (Ovis aries) and goat (Capra hircus) skulls. Archives of Biological Sciences. 2013; 65(1):133-41. [DOI:10.2298/ABS1301141M]
Shawulu JC, Kwari HD, Olopade JO. Morphology of the bones of the skull in the Sahel ecotypes of goats (Capra hircus) in Nigeria. Journal of Animal and Veterinary Advances. 2011; 114(4):1-3. [PMID]
Alsafy MA, El-gendy SA, Abumandour MM. Computed tomography and gross anatomical studies on the head of one-humped camel (Camelus dromedarius). The Anatomical Record. 2014; 297(4):630-42. [DOI:10.1002/ar.22865] [PMID]
de la Barra R, Latorre E, Martínez ME, Calderón C. morphostructural differentiation and variability of merino sheep breed under sustained directional selection. International Journal of Morphology. 2014; 32(3):1069-73. [DOI:10.4067/S0717-95022014000300052]
Todd NE. Qualitative comparison of the cranio-dental osteology of the extant elephants, Elephas Maximus (Asian elephant) and Loxodonta Africana (African elephant). The Anatomical Record: Advances in Integrative Anatomy and Evolutionary Biology. 2010; 293(1):62-73. [DOI:10.1002/ar.21011] [PMID]
Nganvongpanit K, Siengdee P, Buddhachat K, Brown JL, Klinhom S, Pitakarnnop T, et al. Anatomy, histology and elemental profile of long bones and ribs of the Asian elephant (Elephas maximus). Anatomical Science International. 2017; 92(4):554-68. [DOI:10.1007/s12565-016-0361-y] [PMID]
Hidaka S, Matsumoto M, Hiji H, Ohsako S, Nishinakagava H. Morphology and morphometry of skulls of raccoon dogs, Nyctereutes procyonoides and badgers, Meles meles. Journal of Veterinary Medical Science. 1998; 60(2):161-7. [DOI:10.1292/jvms.60.161] [PMID]
Jurgelėnas E. Osteometric analysis of the pelvic bones and sacrum of the red fox and raccoon dog. Veterinarija ir Zootechnika. 2015; 70(92):42-7.
Kieser JA, Groeneveld HT. Mandibulodental allometry in the African wild dog, Lycaon pictus. Journal of Anatomy. 1992; 181(Pt 1):133-7. [PMID] [PMCID]
Pitakarnnop T, Buddhachat K, Euppayo T, Kriangwanich W, Nganvongpanit K. Feline (Felis catus) skull and pelvic morphology and morphometry: Gender-related difference? Anatomia, Histologia, Embryologia. 2017; 46(3):294-303. [DOI:10.1111/ahe.12269] [PMID]
Sicuro FL, Oliveira LF. Skull morphology and functionality of extant Felidae (Mammalia: Carnivora): A phylogenetic and evolutionary perspective. Zoological Journal of the Linnean Society. 2011; 161(2):414-62. [DOI:10.1111/j.1096-3642.2010.00636.x]
Pares-Casanova PM. Reduced skull sexual dimorphism in a local sheep breed. Iranian Journal of Applied Animal Science. 2014; 4(3):643-6.
Kashan NE, Azar GM, Afzalzadeh A, Salehi A. Growth performance and carcass quality of fattening lambs from fat-tailed and tailed sheep breeds. Small Ruminant Research. 2005; 60(3):267-71. [DOI:10.1016/j.smallrumres.2005.01.001]
Marzban Abbasabadi B, Kochakzadeh H, Kaveh Aski A. Evaluating gross anatomy of cervix in Zel sheep. Anatomical Sciences Journal. 2017; 14(3):115-20.
Sarma K. Morphological and craniometrical studies on the skull of Kagani goat (Capra hircus) of Jammu Region/Estudios morfologico y craniometrico de la cabra Kagani (Capra hircus) de la Region de Jammu. International Journal of Morphology. 2006; 24(3):449-56. [DOI:10.4067/S0717-95022006000400025]
Monfared AL, Naji H, Sheibani MT. Applied anatomy of the head region of the Iranian native goats (Capra hircus). Global Veternaria. 2013; 10(1):60-4. [DOI:10.5829/idosi.gv.2013.10.1.71133]
Saber AS, Caceci T, Gummow B, Johns K. Morphometric Studies on the Skull of the Australian Domestic Cat (F. catus) and its Clini-cal Implications for Regional Anesthesia. Journal of Veterinary Anatomy. 2016; 9(1):1-24.
Saber AS, Gummow B. Skull morphometry of the lion (Panthera leo), dog (Canis lupus familiaris) and cat (Felis catus). Journal of Veterinary Anatomy. 2015; 8(1):13-30.[DOI:10.21608/JVA.2015.44849]
JASLOW CR. Sexual dimorphism of cranial suture complexity in wild sheep (Ovis orientalis). Zoological Journal of the Linnean Society. 1989; 95(4):273-84. [DOI:10.1111/j.1096-3642.1989.tb02312.x]
Abramov AV, Tumanov IL. Sexual dimorphism in the skull of the European minkMustela lutreola from NW part of Russia. Acta Theriologica. 2003; 48(2):239-46. [DOI:10.1007/BF03194163]
Farhadinia MS, Kaboli M, Karami M, Farahmand H. Patterns of sexual dimorphism in the Persian Leopard (Panthera pardus saxicolor) and implications for sex differentiation. Zoology in the Middle East. 2014; 60(3):195-207. [DOI:10.1080/09397140.2014.939813]
Parés Casanova PM, Sarma K, Jordana i Vidal J. On biometrical aspects of the cephalic anatomy of Xisqueta sheep (Catalunya, Spain). International Journal of Morphology. 2010; 28(2):347-51. [DOI:10.4067/S0717-95022010000200001]
Özcan S, Aksoy G, Kürtül İ, Aslan K, Özüdoğru Z. A comparative morphometric study on the skull of the Tuj and Morkaraman sheep. Kafkas Üniversitesi Veteriner Fakültesi Dergisi. 2010; 16(1):111-4. [DOI:10.9775/kvfd.2009.518]
Karimi I, Hadipour M, Nikbakht P, Motamedi S. The lower jawbone of Mehreban sheep: A descriptive morphometric approach. World’s Veterinary Journal. 2011; 2:57-60.
Mohamed R, Driscoll M, Mootoo N. Clinical anatomy of the skull of the barbados black belly sheep in trinidad. International Journal of Research in Medical Sciences. 2016; 2(8):8-19.
Type of Study: Original |
Subject:
Morphometry
Received: 2018/12/13 | Accepted: 2019/09/15 | Published: 2020/01/1
Received: 2018/12/13 | Accepted: 2019/09/15 | Published: 2020/01/1
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |