Tue, Jul 1, 2025
Volume 15, Issue 1 (Winter & Spring 2018)
ASJ 2018, 15(1): 31-36 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Rigi Manesh M, Arab M R, Sargolzaei Aval F, Mashhadi M A, Sargolzaei N, Mir M et al . Cisplatin Induce Changes of Cell Surface Glycoconjugates in Germinal Epithelium of Seminiferous Tubules. ASJ 2018; 15 (1) :31-36
URL: http://anatomyjournal.ir/article-1-199-en.html
URL: http://anatomyjournal.ir/article-1-199-en.html
Mohammad Rigi Manesh1 
 , Mohammad Reza Arab1
, Mohammad Reza Arab1 
 , Fereydoon Sargolzaei Aval1
, Fereydoon Sargolzaei Aval1 
 , Mohamad Ali Mashhadi2
, Mohamad Ali Mashhadi2 
 , Narjes Sargolzaei3
, Narjes Sargolzaei3 
 , Mohsen Mir1
, Mohsen Mir1 
 , Masoud Shahraki Salar1
, Masoud Shahraki Salar1 


 , Mohammad Reza Arab1
, Mohammad Reza Arab1 
 , Fereydoon Sargolzaei Aval1
, Fereydoon Sargolzaei Aval1 
 , Mohamad Ali Mashhadi2
, Mohamad Ali Mashhadi2 
 , Narjes Sargolzaei3
, Narjes Sargolzaei3 
 , Mohsen Mir1
, Mohsen Mir1 
 , Masoud Shahraki Salar1
, Masoud Shahraki Salar1 

1- Department of Anatomy, Faculty of Medical Sciences, Zahedan University of Medical Sciences, Zahedan, Iran.
2- Department of Internal Medicine, Faculty of Medical Sciences, Zahedan University of Medical Sciences, Zahedan, Iran.
3- Department of Community Medicine, Faculty of Medical Sciences, Zahedan University of Medical Sciences, Zahedan, Iran.
2- Department of Internal Medicine, Faculty of Medical Sciences, Zahedan University of Medical Sciences, Zahedan, Iran.
3- Department of Community Medicine, Faculty of Medical Sciences, Zahedan University of Medical Sciences, Zahedan, Iran.
Full-Text [PDF 526 kb]
(1535 Downloads)
| Abstract (HTML) (5182 Views)
Full-Text: (2084 Views)
1. Introduction
Cisplatin (CP) is a platinum-based chemotherapeutic drug that effectively used for treatment of different cancers [1] such as ovarian, testes, cervical, bladder, breast, and lung carcinomas [2, 3]. The main mechanism of antineoplastic effect of CP is interfering with purine base of DNA and cessation of cell cycle [4]. Cisplatin create crosslink with cellular DNA of healthy cells of the reproductive, nervous and auditory systems which restricts its use in the treatment of cancer [3, 5].
CP is not a classical DNA-directed alkylating agent but a natural alkylating factor and induce biochemical and tissue changes in testis particularly in germinal epithelium [6]. Testicular injury of CP has been attributed to the direct damage of spermatogenic cell lines [7, 8], Sertoli cells [9], and Leydig cells [10, 11]. Although there are many possible manifestations of toxic damages to spermatogenesis, long-term result of CP administration is sequential testicular atrophy and consequent infertility [9]. Impairment of spermatogenesis [11], chromosomal abnormalities of spermatozoa [12], and transient or permanent azoospermia are the main consequences of CP-based chemotherapy for testicular cancers [13]. Radiotherapy and chemotherapy alter spermatogenesis process. Chemotherapeutic drugs inhibit rapid division of cancer cells as well as other normal cells [14]. Cell surface glycoconjugates as a class of glycolipids or glycoproteins are known to play an important role in cell-cell and cell-extracellular matrix interactions, cell-cell recognition, communication, differentiation, embryogenesis, tumorogenesis, tumor progression, and metastasis [15, 16].
Carbohydrate components of glycoconjugates explain their chemical, physical, structural variety, and biological properties [17]. Most carbohydrates of glycoconjugates are oligo- or poly-saccharides sugars with linear or branched chains, including mannose (Man), fucose (Fuc), galactose (Gal), glucose (Glc), N-acetylgalactosamine (GalNAc), N-acetylglucosamine (GlcNAc) and sialic acid (NeuAc). These monosaccharides in different linear or branched compositions constitute a high diversity in glycoconjugates families, and thus to a high number of possible modifications of proteins and lipids with different properties and functions. Normal structure and function of the cell reflects normal glycosylation pattern of posttranslational modification of cell surface glycoconjugates [18].
Any changes in these terminal sugars determine cells’ behavior in normal and pathologic states such as neoplastic process or metastatic potential of cancer cells [19]. Lectins as group of non-immunogenic biological compounds with plant or animal origin are a valuable marker for determination of terminal sugars of glycoconjugates with high specificity and sensitivity. Lectins detect any changes of glycoconjugates and shows that these molecular modifications are responsible for potential abilities of cancer cells for metastasis and invasion [20, 21].
Glycoconjugates have many important roles in spermatogenesis of mammals. According to Blackmore study glycoconjugates with fucose as a terminal sugar are responsible for vesicular transport from Golgi apparatus to acrosome. It seems that this property may have important roles in cytodifferntiation process of spermiogenesis [22]. Turk et al. reported that CP induces loss of weight of testis, epididymis and seminal vesicle in rats [23]. Bar-Shira et al. showed that CP induces disruption of spermatogenesis by different mechanisms, including apoptosis and break in DNA chains [24]. Seaman et al. believed that toxic effect of CP is mediated by Sertoli cells injury [6]. This study aimed to evaluate the changes of cell surface glycoconjugates in spermatogenic cell lines after cisplatin injection in rats.
2. Material and Methods
In this experimental study, 45 adult male Sprague-Dawley rats weighting 200±30 g were randomly divided into experimental (n=30) and control groups (n=15). Then, they were kept under identical conditions one per cage (12:12 h light: dark cycle, 22°C±2°C and 45%-50% humidity) and fed with standard animal food and water. In the next step, rats of the experimental group were randomly divided into 2 subgroups. Subgroup I (E1) were received 2 consecutive dose of 2.5 mg/kg of cisplatin (MYLAN Co., USA) and experimental subgroup II (E2) received 5 mg/kg of cisplatin. CP was injected intraperitoneally at the beginning of the first and fifth week of the experiment. At the end of the eight week, rats of control and experimental groups were given ether anesthesia and killed, and then their testes were removed, weighted and fixed in 10% formalin saline solution. Tissue samples were processed routinely and 7 µm paraffin sections were stained with H&E method. The sections were examined using light microscope to investigate the spermatogenesis process. After calibration of the light microscope (Motic), histological sections were evaluated for internal and external diameters of seminiferous tubules, epithelial thickness and spermiogenesis index in at least 100 seminiferous tubules in each rat of experimental and control groups.
Lectin histochemistry
Lectin histochemistry was carried out according to the Spicer method [25]. Prepared sections were deparaffinized and hydrated according to routine procedures. Thereafter, the HRP conjugated lectin (PNA [Peanut agglutinin] or UEA [Ulex europaeus agglutinin]) were used, purchased from Sigma Company (USA). For this purpose, the lectins were first diluted with PBS up to 10 μg/mL. Next, the sections were incubated with diluted lectin for two hours in humidified chamber. After this stage, tissue samples were washed with buffer and placed in 0.03% DAB (Diaminobenzidine) as the chromogen solution which contain 0.1% H2O2. The sections were washed, dehydrated, cleared, and mounted. The samples were blindly and separately ranked from 0 (no reaction) to 4 positive (very severe reaction) according to staining intensity.
Statistical analysis
The morphometric data were subjected to parametric methods of analysis of ANOVA followed by Tukey post hoc test (using SPSS version 17.0). Results are presented as mean and standard deviation values. A value of P<0.05 was considered as statistically significant.
3. Results
Statistical data for measured variables showed a significant difference for testis weight between experimental groups and control one (P<0.031) (Table 1). There were also significant decrease in internal and external diameters between experimental groups and control one (P<0.0001) (Table 1). Statistical analysis also demonstrated a significant decrease in epithelial thickness of seminiferous tubules between control and experimental subgroups (P<0.0001) (Table 1). Furthermore, there was a significant decrease in spermiogenesis index in experimental group, compared to the control group (P<0.004, Table 1). Statistical analyses for staining intensity of germinal epithelium to PNA and UEA confirmed a significant difference between control and experimental group (P<0.0001) (Table 1).
Histological findings
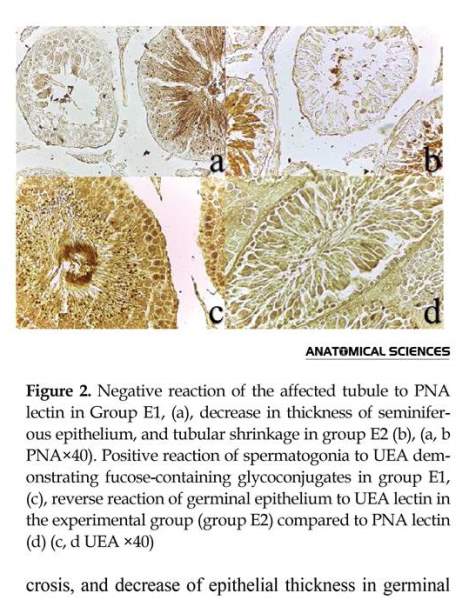
Histological study of the prepared slides showed so many structural changes in the experimental groups compared to the control group. These histological changes are loss of intercellular adhesion between Sertoli and spermatogenic cell lines and a decrease in thickness of germinal epithelium, tubular shrinkage, germ cell degeneration and interstitial edema. Maturation arrest in spermatogenic cell lines and perivascular fibrosis were the most prominent findings in the experimental groups (Figures 1, 2).
4. Discussion
The present study results showed the staining intensity of germinal epithelium to PNA lectin which demonstrate terminal sugar of Gal/GalNac containing glycoconjugates increase from basal to apical compartment in experimental group compared with the control. This glycoconjugates may be very important for cell-cell and cell-extracellular matrix interaction and cytodifferentiation [15, 16]. In germinal epithelium, the proper organization of spermatogenic cell lines is very important for normal spermatogenesis. Our study showed a reverse gradient of staining intensity of germinal epithelium to UEA compared to PNA lectin. Spermatogonia have
Cisplatin (CP) is a platinum-based chemotherapeutic drug that effectively used for treatment of different cancers [1] such as ovarian, testes, cervical, bladder, breast, and lung carcinomas [2, 3]. The main mechanism of antineoplastic effect of CP is interfering with purine base of DNA and cessation of cell cycle [4]. Cisplatin create crosslink with cellular DNA of healthy cells of the reproductive, nervous and auditory systems which restricts its use in the treatment of cancer [3, 5].
CP is not a classical DNA-directed alkylating agent but a natural alkylating factor and induce biochemical and tissue changes in testis particularly in germinal epithelium [6]. Testicular injury of CP has been attributed to the direct damage of spermatogenic cell lines [7, 8], Sertoli cells [9], and Leydig cells [10, 11]. Although there are many possible manifestations of toxic damages to spermatogenesis, long-term result of CP administration is sequential testicular atrophy and consequent infertility [9]. Impairment of spermatogenesis [11], chromosomal abnormalities of spermatozoa [12], and transient or permanent azoospermia are the main consequences of CP-based chemotherapy for testicular cancers [13]. Radiotherapy and chemotherapy alter spermatogenesis process. Chemotherapeutic drugs inhibit rapid division of cancer cells as well as other normal cells [14]. Cell surface glycoconjugates as a class of glycolipids or glycoproteins are known to play an important role in cell-cell and cell-extracellular matrix interactions, cell-cell recognition, communication, differentiation, embryogenesis, tumorogenesis, tumor progression, and metastasis [15, 16].
Carbohydrate components of glycoconjugates explain their chemical, physical, structural variety, and biological properties [17]. Most carbohydrates of glycoconjugates are oligo- or poly-saccharides sugars with linear or branched chains, including mannose (Man), fucose (Fuc), galactose (Gal), glucose (Glc), N-acetylgalactosamine (GalNAc), N-acetylglucosamine (GlcNAc) and sialic acid (NeuAc). These monosaccharides in different linear or branched compositions constitute a high diversity in glycoconjugates families, and thus to a high number of possible modifications of proteins and lipids with different properties and functions. Normal structure and function of the cell reflects normal glycosylation pattern of posttranslational modification of cell surface glycoconjugates [18].
Any changes in these terminal sugars determine cells’ behavior in normal and pathologic states such as neoplastic process or metastatic potential of cancer cells [19]. Lectins as group of non-immunogenic biological compounds with plant or animal origin are a valuable marker for determination of terminal sugars of glycoconjugates with high specificity and sensitivity. Lectins detect any changes of glycoconjugates and shows that these molecular modifications are responsible for potential abilities of cancer cells for metastasis and invasion [20, 21].
Glycoconjugates have many important roles in spermatogenesis of mammals. According to Blackmore study glycoconjugates with fucose as a terminal sugar are responsible for vesicular transport from Golgi apparatus to acrosome. It seems that this property may have important roles in cytodifferntiation process of spermiogenesis [22]. Turk et al. reported that CP induces loss of weight of testis, epididymis and seminal vesicle in rats [23]. Bar-Shira et al. showed that CP induces disruption of spermatogenesis by different mechanisms, including apoptosis and break in DNA chains [24]. Seaman et al. believed that toxic effect of CP is mediated by Sertoli cells injury [6]. This study aimed to evaluate the changes of cell surface glycoconjugates in spermatogenic cell lines after cisplatin injection in rats.
2. Material and Methods
In this experimental study, 45 adult male Sprague-Dawley rats weighting 200±30 g were randomly divided into experimental (n=30) and control groups (n=15). Then, they were kept under identical conditions one per cage (12:12 h light: dark cycle, 22°C±2°C and 45%-50% humidity) and fed with standard animal food and water. In the next step, rats of the experimental group were randomly divided into 2 subgroups. Subgroup I (E1) were received 2 consecutive dose of 2.5 mg/kg of cisplatin (MYLAN Co., USA) and experimental subgroup II (E2) received 5 mg/kg of cisplatin. CP was injected intraperitoneally at the beginning of the first and fifth week of the experiment. At the end of the eight week, rats of control and experimental groups were given ether anesthesia and killed, and then their testes were removed, weighted and fixed in 10% formalin saline solution. Tissue samples were processed routinely and 7 µm paraffin sections were stained with H&E method. The sections were examined using light microscope to investigate the spermatogenesis process. After calibration of the light microscope (Motic), histological sections were evaluated for internal and external diameters of seminiferous tubules, epithelial thickness and spermiogenesis index in at least 100 seminiferous tubules in each rat of experimental and control groups.
Lectin histochemistry
Lectin histochemistry was carried out according to the Spicer method [25]. Prepared sections were deparaffinized and hydrated according to routine procedures. Thereafter, the HRP conjugated lectin (PNA [Peanut agglutinin] or UEA [Ulex europaeus agglutinin]) were used, purchased from Sigma Company (USA). For this purpose, the lectins were first diluted with PBS up to 10 μg/mL. Next, the sections were incubated with diluted lectin for two hours in humidified chamber. After this stage, tissue samples were washed with buffer and placed in 0.03% DAB (Diaminobenzidine) as the chromogen solution which contain 0.1% H2O2. The sections were washed, dehydrated, cleared, and mounted. The samples were blindly and separately ranked from 0 (no reaction) to 4 positive (very severe reaction) according to staining intensity.
Statistical analysis
The morphometric data were subjected to parametric methods of analysis of ANOVA followed by Tukey post hoc test (using SPSS version 17.0). Results are presented as mean and standard deviation values. A value of P<0.05 was considered as statistically significant.
3. Results
Statistical data for measured variables showed a significant difference for testis weight between experimental groups and control one (P<0.031) (Table 1). There were also significant decrease in internal and external diameters between experimental groups and control one (P<0.0001) (Table 1). Statistical analysis also demonstrated a significant decrease in epithelial thickness of seminiferous tubules between control and experimental subgroups (P<0.0001) (Table 1). Furthermore, there was a significant decrease in spermiogenesis index in experimental group, compared to the control group (P<0.004, Table 1). Statistical analyses for staining intensity of germinal epithelium to PNA and UEA confirmed a significant difference between control and experimental group (P<0.0001) (Table 1).
Histological findings
Histological study of the prepared slides showed so many structural changes in the experimental groups compared to the control group. These histological changes are loss of intercellular adhesion between Sertoli and spermatogenic cell lines and a decrease in thickness of germinal epithelium, tubular shrinkage, germ cell degeneration and interstitial edema. Maturation arrest in spermatogenic cell lines and perivascular fibrosis were the most prominent findings in the experimental groups (Figures 1, 2).
4. Discussion
The present study results showed the staining intensity of germinal epithelium to PNA lectin which demonstrate terminal sugar of Gal/GalNac containing glycoconjugates increase from basal to apical compartment in experimental group compared with the control. This glycoconjugates may be very important for cell-cell and cell-extracellular matrix interaction and cytodifferentiation [15, 16]. In germinal epithelium, the proper organization of spermatogenic cell lines is very important for normal spermatogenesis. Our study showed a reverse gradient of staining intensity of germinal epithelium to UEA compared to PNA lectin. Spermatogonia have
no reaction to PNA; spermatozoa’s tail have a severe staining intensity to this lectin. The study of Sato et al. showed similar results for germinal epithelium and also confirmed lamina propria have a positive reaction to PNA lectin [26]. Furthermore, results of the present study showed the staining intensity of germinal epithelium to UEA lectin which demonstrate terminal sugar of fucose-containing glycoconjugates increases from apical to basal compartment in the experimental groups compared with the control group. Spermatogonia have a high affinity to UEA lectin, but spermatozoa’s tail has a mild reaction. The study of Blackmore et al. showed that fucose-containing glycoconjugates facilitate cell-cell interactions [22] suggesting the main role for this fucosylated carbohydrates in spermiogenesis in cancer patients. Degeneration of germinal epithelium of seminiferous tubules may induce changes in expression pattern of glycoconjugates after cisplatin injection. This changes including germ cells apoptosis and loss of junction between spermatogenic cell lines and sertoli cells [6, 20].
The present study shows that cisplatin is able to change different parameters in testis such as decrease in testis weight between experimental and control groups. The study of Cherry et al. showed that cisplatin injection induces decrease in testis weight, sperm count and loss of germ cells in germinal epithelium [11] which is accordance with our results. Thus, the decrease in testis weight could be a manifestation of degeneration, necrosis, and decrease of epithelial thickness in germinal epithelium [27].
The present study results showed significant decrease in internal and external diameters of seminiferous tubules after cisplatin injection which agrees with the study of Leandra and associates [2]. Loss of cell junctions may be a manifestation of spermatogenesis impairment [28]. The junctional complex between Sertoli and spermatogenic cell lines is necessary for preservation of morphology and volume of tubules [29]. The study of He et al. showed that vimentin plays an important role in cell junctions between adjacent Sertoli cells [30], which its expression decreases after cisplatin injection [31], a possible mechanism for changes in diameters of seminiferous tubules. Thus, the diameter of seminiferous tubules is one of most important parameters for evaluation of spermatogenesis [32]. On the other hand, the thickness of germinal epithelium is another good marker for the evaluation of spermatogenesis. Chemotherapeutic drugs induce damage to germinal epithelium which may lead to oligospermia or azoospermia in cancer patients [11].
Statistical analysis of the present study showed decrease in epithelial thickness of seminiferous tubules between experimental and control groups. Also qualitative result of the present study showed tubular shrinkage and interstitial edema in experimental group which are in accordance with Beytur et al. study results [33]. Furthermore, our results showed spermiogenesis index significantly decreased in experimental groups compared with the control group. In accordance with our results, Rezvanfar et al. study showed that sperm count, spermiogenesis index and differentiated germ cells decreased after cisplatin injection [34]. It seems that cisplatin induces many quantitative and qualitative changes in rats’ testes in a dose dependent manner, including extensive changes in distribution pattern of fucose- and Gal/GalNac-containing glycoconjugates in seminiferous epithelium, loss of intercellular junction between Sertoli cells, decrease in the height of germinal epithelium, tubular shrinkage, germ cell degeneration, interstitial edema, perivascular fibrosis, decrease in internal and external diameters of seminiferous tubule, decrease in germinal epithelial thickness and spermiogenesis index.
Acknowledgments
The authors wish to thank the Deputy for Research of Zahedan University of Medical Sciences for the financial supports (IR.ZAUMS.REC.1394.352).
Conflict of Interest
The authors declared no conflicts of interest.
References
The present study shows that cisplatin is able to change different parameters in testis such as decrease in testis weight between experimental and control groups. The study of Cherry et al. showed that cisplatin injection induces decrease in testis weight, sperm count and loss of germ cells in germinal epithelium [11] which is accordance with our results. Thus, the decrease in testis weight could be a manifestation of degeneration, necrosis, and decrease of epithelial thickness in germinal epithelium [27].
The present study results showed significant decrease in internal and external diameters of seminiferous tubules after cisplatin injection which agrees with the study of Leandra and associates [2]. Loss of cell junctions may be a manifestation of spermatogenesis impairment [28]. The junctional complex between Sertoli and spermatogenic cell lines is necessary for preservation of morphology and volume of tubules [29]. The study of He et al. showed that vimentin plays an important role in cell junctions between adjacent Sertoli cells [30], which its expression decreases after cisplatin injection [31], a possible mechanism for changes in diameters of seminiferous tubules. Thus, the diameter of seminiferous tubules is one of most important parameters for evaluation of spermatogenesis [32]. On the other hand, the thickness of germinal epithelium is another good marker for the evaluation of spermatogenesis. Chemotherapeutic drugs induce damage to germinal epithelium which may lead to oligospermia or azoospermia in cancer patients [11].
Statistical analysis of the present study showed decrease in epithelial thickness of seminiferous tubules between experimental and control groups. Also qualitative result of the present study showed tubular shrinkage and interstitial edema in experimental group which are in accordance with Beytur et al. study results [33]. Furthermore, our results showed spermiogenesis index significantly decreased in experimental groups compared with the control group. In accordance with our results, Rezvanfar et al. study showed that sperm count, spermiogenesis index and differentiated germ cells decreased after cisplatin injection [34]. It seems that cisplatin induces many quantitative and qualitative changes in rats’ testes in a dose dependent manner, including extensive changes in distribution pattern of fucose- and Gal/GalNac-containing glycoconjugates in seminiferous epithelium, loss of intercellular junction between Sertoli cells, decrease in the height of germinal epithelium, tubular shrinkage, germ cell degeneration, interstitial edema, perivascular fibrosis, decrease in internal and external diameters of seminiferous tubule, decrease in germinal epithelial thickness and spermiogenesis index.
Acknowledgments
The authors wish to thank the Deputy for Research of Zahedan University of Medical Sciences for the financial supports (IR.ZAUMS.REC.1394.352).
Conflict of Interest
The authors declared no conflicts of interest.
References
- Jordan P, Carmo-Fonseca M. Molecular mechanisms involved in cisplatin cytotoxicity. Cellular and Molecular Life Sciences. 2000; 57(8):1229–35. doi: 10.1007/pl00000762
- Lirdi LC, Stumpp T, Sasso Cerri E, Miraglia SM. Amifostine protective effect on cisplatin-treated rat testis. The Anatomical Record: Advances in Integrative Anatomy and Evolutionary Biology. 2008; 291(7):797–808. doi: 10.1002/ar.20693
- Ahmed EA, Omar HM, elghaffar SKA, Ragb SMM, Nasser AY. The antioxidant activity of Vitamin C, DPPD and l-cysteine against Cisplatin-induced testicular oxidative damage in rats. Food and Chemical Toxicology. 2011; 49(5):1115–21. doi: 10.1016/j.fct.2011.02.002
- Diaz R, Jorda MV, Reynes G, Aparicio J, Segura A, Amador R, et al. Neoadjuvant cisplatin and etoposide, with or without tamoxifen, prior to radiotherapy in high-grade gliomas: A single-center experience. Anti-Cancer Drugs. 2005; 16(3):323–9. doi: 10.1097/00001813-200503000-00012
- Ilbey YO, Ozbek E, Cekmen M, Simsek A, Otunctemur A, Somay A. Protective effect of curcumin in cisplatin-induced oxidative injury in rat testis: mitogen-activated protein kinase and nuclear factor-kappa B signaling pathways. Human Reproduction. 2009; 24(7):1717–25. doi: 10.1093/humrep/dep058
- Seaman F, Sawhney P, Giammona CJ, Richburg JH. Cisplatin-induced pulse of germ cell apoptosis precedes long-term elevated apoptotic rates in C57/BL/6 mouse testis. Apoptosis. 2003; 8(1):101–8. doi: 10.1023/a:1021734604913
- Aminsharifi AR, Talaei T, Kumar V, Sabayan B, Samani S, Mohamadhoseini E. A postulated role of testosterone for prevention of cisplatin gonadal toxicity. Medical Hypotheses. 2007; 68(3):525–7. doi: 10.1016/j.mehy.2006.08.023
- Meistrich ML, Finch M, da Cunha MF, Hacker U, Au WW. Damaging effects of fourteen chemotherapeutic drugs on mouse testis cells. Cancer Research. 1982; 42(1):122-31. PMID: 7198505
- Boekelheide K. Mechanisms of toxic damage to spermatogenesis. Journal of the National Cancer Institute Monographs. 2005; 2005(34):6–8. doi: 10.1093/jncimonographs/lgi006
- Vawda AI. Effect of testosterone on cisplatin-induced testicular damage. Archives of Andrology. 1994; 32(1):53–7. doi: 10.3109/01485019408987767
- Cherry SM, Hunt PA, Hassold TJ. Cisplatin disrupts mammalian spermatogenesis, but does not affect recombination or chromosome segregation. Mutation Research/Genetic Toxicology and Environmental Mutagenesis. 2004; 564(2):115–28. doi: 10.1016/j.mrgentox.2004.08.010
- Martin RH, Ernst S, Rademaker A, Barclay L, Ko E, Summers N. Analysis of sperm chromosome complements before, during, and after chemotherapy. Cancer Genetics and Cytogenetics. 1999; 108(2):133–6. doi: 10.1016/s0165-4608(98)00125-3
- Howell SJ. Spermatogenesis after cancer treatment: Damage and recovery. Journal of the National Cancer Institute Monographs. 2005; 2005(34):12–7. doi: 10.1093/jncimonographs/lgi003
- Rad JS, Nejad DM, Roshankar L, Karimipor M, Ghanbari AA, Aazami A, et al. A study on the effect of thiotepa on mice spermatogenesis using light and electronic microscope. Pakistan Journal of Biological Sciences. 2008; 11(15):1929–34. doi: 10.3923/pjbs.2008.1929.1934
- Xu X-C, Brinck U, Schauer A, Gabius HJ. Differential binding activities of lectins and neoglycoproteins in human testicular tumors. Urological Research. 2000; 28(1):62–8. doi: 10.1007/s002400050012
- Thies A, Moll I, Berger J, Schumacher U. Lectin binding to cutaneous malignant melanoma: HPA is associated with metastasis formation. British Journal of Cancer. 2001; 84(6):819–23. doi: 10.1054/bjoc.2000.1673.
- Davis GB. Recent developments in glycoconjugates. Journal of the Chemical Society, Perkin Transactions 1. 1999; (22):3215. doi: 10.1039/a809773i
- Wang PH. Altered glycosylation in cancer: Sialic acids and sialyltransferases. Journal of Cancer Molecules. 2005; 1(2):73-81.
- Arab MR, Salari S, Karimi M, Mofidpour H. Lectin histochemical study of cell surface glycoconjugate in gastric carcinoma using helix pomatia agglutinin. Acta Medica Iranica. 2010; 48(4):209.
- Krogerus L, Andersson LC. Different lectin-binding patterns in primary breast cancers and their metastases. Cancer. 1990; 66(8):1802-9. doi: 10.1002/1097-0142(19901015)66:8<1802::aid-cncr2820660827>3.0.co; 2-z
- Sherwani AF, Mohmood S, Khan F, Khan RH, Azfer MA. Characterization of lectins and their specificity in carcinomas: An appraisal. Indian Journal of Clinical Biochemistry. 2003; 18(2):169–80. doi: 10.1007/bf02867384
- Blackmore PF. The neoglycoprotein mannosebovine serum albumin, but not progesterone, activates T-type calcium channels in human spermatozoa. Molecular Human Reproduction. 1999; 5(6):498–506. doi: 10.1093/molehr/5.6.498
- Türk G, Ateşşahin A, Sönmez M, Yüce A, Çeribaşi AO. Lycopene protects against cyclosporine A-induced testicular toxicity in rats. Theriogenology. 2007; 67(4):778–85. doi: 10.1016/j.theriogenology.2006.10.013
- Bar Shira Maymon B, Yogev L, Marks A, Hauser R, Botchan A, Yavetz H. Sertoli cell inactivation by cytotoxic damage to the human testis after cancer chemotherapy. Fertility and Sterility. 2004; 81(5):1391–4. doi: 10.1016/j.fertnstert.2003.09.078
- Spicer SS, Schulte BA. Detection and differentiation of glycoconjugates in various cell types by lectin histochemistry. Basic and Applied Histochemistry. 1988; 32(3):307-20. PMID:3067702
- Sato Y, Nozawa S, Yoshiike M, Otoi T, Iwamoto T. Glycoconjugates recognized by peanut agglutinin lectin in the inner acellular layer of the lamina propria of seminiferous tubules in human testes showing impaired spermatogenesis. Human Reproduction. 2012; 27(3):659–68. doi: 10.1093/humrep/der430
- Ilbey YO, Ozbek E, Simsek A, Otunctemur A, Cekmen M, Somay A. Potential chemoprotective effect of melatonin in cyclophosphamide- and cisplatin-induced testicular damage in rats. Fertility and Sterility. 2009; 92(3):1124–32. doi: 10.1016/j.fertnstert.2008.07.1758
- Bouchard MJ, Dong Y, McDermott BM, Lam D-H, Brown KR, Shelanski M, et al. Defects in nuclear and cytoskeletal morphology and mitochondrial localization in spermatozoa of mice lacking nectin-2, a component of cell-cell adherens junctions. Molecular and Cellular Biology. 2000; 20(8):2865–73. doi: 10.1128/mcb.20.8.2865-2873.2000
- Mruk DD, Cheng CY. Sertoli-sertoli and sertoli-germ cell interactions and their significance in germ cell movement in the seminiferous epithelium during spermatogenesis. Endocrine Reviews. 2004; 25(5):747–806. doi: 10.1210/er.2003-0022
- He D, Zhang D, Wei G, Lin T, Li X. Cytoskeleton vimentin disruption of mouse sertoli cells injured by nitrogen mustard in vitro. Journal of Andrology. 2006; 28(3):389–96. doi: 10.2164/jandrol.106.000455
- Liu Z, Sun Y, Su L, Sun Y, Kong S, Chang X, et al. Effects of cisplatin on testicular enzymes and Sertoli cell function in rats. Fundamental Toxicological Sciences. 2015; 2(4):137–45. doi: 10.2131/fts.2.137
- França LR, Russell LD. The testis of domestic animals. In F Martinez-Garcia, J Regadera (Eds.), Male Reproduction: A Multidisciplinary Overview. Madrid: Churchill Communications; 1998.
- Beytur A, Ciftci O, Oguz F, Oguzturk H, Yılmaz F. Montelukast attenuates side effects of cisplatin including testicular, spermatological, and hormonal damage in male rats. Cancer Chemotherapy and Pharmacology. 2011; 69(1):207–13. doi: 10.1007/s00280-011-1692-y
- Rezvanfar MA, Rezvanfar MA, Shahverdi AR, Ahmadi A, Baeeri M, Mohammadirad A, et al. Protection of cisplatin-induced spermatotoxicity, DNA damage and chromatin abnormality by selenium nano-particles. Toxicology and Applied Pharmacology. 2013; 266(3):356–65. doi: 10.1016/j.taap.2012.11.025
Type of Study: Original |
Subject:
Histology
Received: 2016/07/12 | Accepted: 2017/11/15 | Published: 2018/01/1
Received: 2016/07/12 | Accepted: 2017/11/15 | Published: 2018/01/1
References
1. Jordan P, Carmo-Fonseca M. Molecular mechanisms involved in cisplatin cytotoxicity. Cellular and Molecular Life Sciences. 2000; 57(8):1229–35. doi: 10.1007/pl00000762 [DOI:10.1007/PL00000762]
2. Lirdi LC, Stumpp T, Sasso Cerri E, Miraglia SM. Amifostine protective effect on cisplatin-treated rat testis. The Anatomical Record: Advances in Integrative Anatomy and Evolutionary Biology. 2008; 291(7):797–808. doi: 10.1002/ar.20693 [DOI:10.1002/ar.20693]
3. Ahmed EA, Omar HM, elghaffar SKA, Ragb SMM, Nasser AY. The antioxidant activity of Vitamin C, DPPD and l-cysteine against Cisplatin-induced testicular oxidative damage in rats. Food and Chemical Toxicology. 2011; 49(5):1115–21. doi: 10.1016/j.fct.2011.02.002 [DOI:10.1016/j.fct.2011.02.002]
4. Diaz R, Jorda MV, Reynes G, Aparicio J, Segura A, Amador R, et al. Neoadjuvant cisplatin and etoposide, with or without tamoxifen, prior to radiotherapy in high-grade gliomas: A single-center experience. Anti-Cancer Drugs. 2005; 16(3):323–9. doi: 10.1097/00001813-200503000-00012 [DOI:10.1097/00001813-200503000-00012]
5. Ilbey YO, Ozbek E, Cekmen M, Simsek A, Otunctemur A, Somay A. Protective effect of curcumin in cisplatin-induced oxidative injury in rat testis: mitogen-activated protein kinase and nuclear factor-kappa B signaling pathways. Human Reproduction. 2009; 24(7):1717–25. doi: 10.1093/humrep/dep058 [DOI:10.1093/humrep/dep058]
6. Seaman F, Sawhney P, Giammona CJ, Richburg JH. Cisplatin-induced pulse of germ cell apoptosis precedes long-term elevated apoptotic rates in C57/BL/6 mouse testis. Apoptosis. 2003; 8(1):101–8. doi: 10.1023/a:1021734604913 [DOI:10.1023/A:1021734604913]
7. Aminsharifi AR, Talaei T, Kumar V, Sabayan B, Samani S, Mohamadhoseini E. A postulated role of testosterone for prevention of cisplatin gonadal toxicity. Medical Hypotheses. 2007; 68(3):525–7. doi: 10.1016/j.mehy.2006.08.023 [DOI:10.1016/j.mehy.2006.08.023]
8. Meistrich ML, Finch M, da Cunha MF, Hacker U, Au WW. Damaging effects of fourteen chemotherapeutic drugs on mouse testis cells. Cancer Research. 1982; 42(1):122-31. PMID: 7198505 [PMID]
9. Boekelheide K. Mechanisms of toxic damage to spermatogenesis. Journal of the National Cancer Institute Monographs. 2005; 2005(34):6–8. doi: 10.1093/jncimonographs/lgi006 [DOI:10.1093/jncimonographs/lgi006]
10. Vawda AI. Effect of testosterone on cisplatin-induced testicular damage. Archives of Andrology. 1994; 32(1):53–7. doi: 10.3109/01485019408987767 [DOI:10.3109/01485019408987767]
11. Cherry SM, Hunt PA, Hassold TJ. Cisplatin disrupts mammalian spermatogenesis, but does not affect recombination or chromosome segregation. Mutation Research/Genetic Toxicology and Environmental Mutagenesis. 2004; 564(2):115–28. doi: 10.1016/j.mrgentox.2004.08.010 [DOI:10.1016/j.mrgentox.2004.08.010]
12. Martin RH, Ernst S, Rademaker A, Barclay L, Ko E, Summers N. Analysis of sperm chromosome complements before, during, and after chemotherapy. Cancer Genetics and Cytogenetics. 1999; 108(2):133–6. doi: 10.1016/s0165-4608(98)00125-3 [DOI:10.1016/S0165-4608(98)00125-3]
13. Howell SJ. Spermatogenesis after cancer treatment: Damage and recovery. Journal of the National Cancer Institute Monographs. 2005; 2005(34):12–7. doi: 10.1093/jncimonographs/lgi003 [DOI:10.1093/jncimonographs/lgi003]
14. Rad JS, Nejad DM, Roshankar L, Karimipor M, Ghanbari AA, Aazami A, et al. A study on the effect of thiotepa on mice spermatogenesis using light and electronic microscope. Pakistan Journal of Biological Sciences. 2008; 11(15):1929–34. doi: 10.3923/pjbs.2008.1929.1934 [DOI:10.3923/pjbs.2008.1929.1934]
15. Xu X-C, Brinck U, Schauer A, Gabius HJ. Differential binding activities of lectins and neoglycoproteins in human testicular tumors. Urological Research. 2000; 28(1):62–8. doi: 10.1007/s002400050012 [DOI:10.1007/s002400050012]
16. Thies A, Moll I, Berger J, Schumacher U. Lectin binding to cutaneous malignant melanoma: HPA is associated with metastasis formation. British Journal of Cancer. 2001; 84(6):819–23. doi: 10.1054/bjoc.2000.1673. [DOI:10.1054/bjoc.2000.1673]
17. Davis GB. Recent developments in glycoconjugates. Journal of the Chemical Society, Perkin Transactions 1. 1999; (22):3215. doi: 10.1039/a809773i [DOI:10.1039/a809773i]
18. Wang PH. Altered glycosylation in cancer: Sialic acids and sialyltransferases. Journal of Cancer Molecules. 2005; 1(2):73-81.
19. Arab MR, Salari S, Karimi M, Mofidpour H. Lectin histochemical study of cell surface glycoconjugate in gastric carcinoma using helix pomatia agglutinin. Acta Medica Iranica. 2010; 48(4):209. [PMID]
20. Krogerus L, Andersson LC. Different lectin-binding patterns in primary breast cancers and their metastases. Cancer. 1990; 66(8):1802-9. doi: 10.1002/1097-0142(19901015)66:8<1802::aid-cncr2820660827>3.0.co; 2-z
21. Sherwani AF, Mohmood S, Khan F, Khan RH, Azfer MA. Characterization of lectins and their specificity in carcinomas: An appraisal. Indian Journal of Clinical Biochemistry. 2003; 18(2):169–80. doi: 10.1007/bf02867384 [DOI:10.1007/BF02867384]
22. Blackmore PF. The neoglycoprotein mannosebovine serum albumin, but not progesterone, activates T-type calcium channels in human spermatozoa. Molecular Human Reproduction. 1999; 5(6):498–506. doi: 10.1093/molehr/5.6.498 [DOI:10.1093/molehr/5.6.498]
23. Türk G, Ateşşahin A, Sönmez M, Yüce A, Çeribaşi AO. Lycopene protects against cyclosporine A-induced testicular toxicity in rats. Theriogenology. 2007; 67(4):778–85. doi: 10.1016/j.theriogenology.2006.10.013 [DOI:10.1016/j.theriogenology.2006.10.013]
24. Bar Shira Maymon B, Yogev L, Marks A, Hauser R, Botchan A, Yavetz H. Sertoli cell inactivation by cytotoxic damage to the human testis after cancer chemotherapy. Fertility and Sterility. 2004; 81(5):1391–4. doi: 10.1016/j.fertnstert.2003.09.078 [DOI:10.1016/j.fertnstert.2003.09.078]
25. Spicer SS, Schulte BA. Detection and differentiation of glycoconjugates in various cell types by lectin histochemistry. Basic and Applied Histochemistry. 1988; 32(3):307-20. PMID:3067702 [PMID]
26. Sato Y, Nozawa S, Yoshiike M, Otoi T, Iwamoto T. Glycoconjugates recognized by peanut agglutinin lectin in the inner acellular layer of the lamina propria of seminiferous tubules in human testes showing impaired spermatogenesis. Human Reproduction. 2012; 27(3):659–68. doi: 10.1093/humrep/der430 [DOI:10.1093/humrep/der430]
27. Ilbey YO, Ozbek E, Simsek A, Otunctemur A, Cekmen M, Somay A. Potential chemoprotective effect of melatonin in cyclophosphamide- and cisplatin-induced testicular damage in rats. Fertility and Sterility. 2009; 92(3):1124–32. doi: 10.1016/j.fertnstert.2008.07.1758 [DOI:10.1016/j.fertnstert.2008.07.1758]
28. Bouchard MJ, Dong Y, McDermott BM, Lam D-H, Brown KR, Shelanski M, et al. Defects in nuclear and cytoskeletal morphology and mitochondrial localization in spermatozoa of mice lacking nectin-2, a component of cell-cell adherens junctions. Molecular and Cellular Biology. 2000; 20(8):2865–73. doi: 10.1128/mcb.20.8.2865-2873.2000 [DOI:10.1128/MCB.20.8.2865-2873.2000]
29. Mruk DD, Cheng CY. Sertoli-sertoli and sertoli-germ cell interactions and their significance in germ cell movement in the seminiferous epithelium during spermatogenesis. Endocrine Reviews. 2004; 25(5):747–806. doi: 10.1210/er.2003-0022 [DOI:10.1210/er.2003-0022]
30. He D, Zhang D, Wei G, Lin T, Li X. Cytoskeleton vimentin disruption of mouse sertoli cells injured by nitrogen mustard in vitro. Journal of Andrology. 2006; 28(3):389–96. doi: 10.2164/jandrol.106.000455 [DOI:10.2164/jandrol.106.000455]
31. Liu Z, Sun Y, Su L, Sun Y, Kong S, Chang X, et al. Effects of cisplatin on testicular enzymes and Sertoli cell function in rats. Fundamental Toxicological Sciences. 2015; 2(4):137–45. doi: 10.2131/fts.2.137 [DOI:10.2131/fts.2.137]
32. França LR, Russell LD. The testis of domestic animals. In F Martinez-Garcia, J Regadera (Eds.), Male Reproduction: A Multidisciplinary Overview. Madrid: Churchill Communications; 1998.
33. Beytur A, Ciftci O, Oguz F, Oguzturk H, Yılmaz F. Montelukast attenuates side effects of cisplatin including testicular, spermatological, and hormonal damage in male rats. Cancer Chemotherapy and Pharmacology. 2011; 69(1):207–13. doi: 10.1007/s00280-011-1692-y [DOI:10.1007/s00280-011-1692-y]
34. Rezvanfar MA, Rezvanfar MA, Shahverdi AR, Ahmadi A, Baeeri M, Mohammadirad A, et al. Protection of cisplatin-induced spermatotoxicity, DNA damage and chromatin abnormality by selenium nano-particles. Toxicology and Applied Pharmacology. 2013; 266(3):356–65. doi: 10.1016/j.taap.2012.11.025 [DOI:10.1016/j.taap.2012.11.025]
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |


.jpg)