Sat, Jul 12, 2025
Volume 15, Issue 2 (Summer & Autumn 2018)
ASJ 2018, 15(2): 41-46 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Mosavian Naeini R, Sahebalzamani M, Nazem M N. Histomorphometrical Changes on the Knee Joints of Male and Female Rats After Moderate Exercise Program. ASJ 2018; 15 (2) :41-46
URL: http://anatomyjournal.ir/article-1-184-en.html
URL: http://anatomyjournal.ir/article-1-184-en.html
1- Department of Sport Medicine, Faculty of Physical Education, Shahid Bahonar University of Kerman, Kerman, Iran.
2- Department of Basic Sciences, Faculty of Veterinary Medicine, Shahid Bahonar University of Kerman, Kerman, Iran.
2- Department of Basic Sciences, Faculty of Veterinary Medicine, Shahid Bahonar University of Kerman, Kerman, Iran.
Full-Text [PDF 522 kb]
(1416 Downloads)
| Abstract (HTML) (5343 Views)
Our obtained results revealed that females show more cartilage changes in a moderate exercise compared to males with the same body weight. Some studies indicate that the knee cartilage volume differences are noticeable in adults aged between 26 and 61 years. Based on this study, radiographic assessments revealed that men’s cartilage volume is 33%–42% greater than that in women in normal cases [26]. Our study revealed that male rats had at least about 35% greater cartilage volume than females in the control groups. In contrast with our results, Faber et al. [27] reported that the volume of knee cartilage is more than that in the females. They believed that sex differences didn’t significantly affect the cartilage volume after matching body weight and height. Ding et al. reported that body and bone size have significant effect on this difference, which is in agreement with some ideas that cartilage volume has a direct relationship with bone size [28-30]. In other words, males have a thicker cartilage than females.
In our study, we showed some significant changes in the same body weight and age. However, physical activity measurements and fitness did not account for the detected differences. The differences that related to the sex declined but continued at all parts in the whole of the joint significantly. It suggests that other parameters may explain the sex differences. This may be because of limitations in bone measurements. However, these measures are highly reproducible, best reflect the surface and structure of the joint. Therefore, the growth factors and sex hormones are the most important candidates to explain this difference. Presence of the receptors of progesterone, estrogen and testosterone – some of the sex hormones – are reported in the human fetal cartilaginous tissue [31]. It has been shown that androgens secretion can stimulate the proliferation of chondrocytes. These cells produce and release the collagen and proteoglycans that are necessary to conserve and sustain the cartilage [32]. It has been shown that hormone therapy in post-menopausal women leads to increase in the cartilage volume [33].
Some research studies revealed that blood serum testosterone has a positive direct effect on the cartilage volume of the knee joint at all sites [34]. Also growth factors such as transforming growth factor-ɞ and insulin-like growth factor-1 have important effect in development of the articular cartilage and proteoglycan production [35]. It is noticeable that evaluating the effect of sex hormone and growth factors on the cartilage formation and its volume needs more investigation. Our results showed significant changes in the knee cartilage after a moderate exercise in male and female rats with similar age and weight. It also suggests that the exercise program in females should be different from that in males.
Ethical Considerations
Compliance with ethical guidelines
All steps of this study were approved by the Animal Ethics Committee, based on the Research Council of the Veterinary School, Shahid Bahonar University of Kerman, Iran.
Funding
This research was financially supported by the Research Council of Shahid Bahonar University of Kerman (No: 1.7.1395).
Conflict of interest
The authors declared no conflict of interest.
Acknowledgements
The authors would like to thank Mr. Mazhab Jaafari for providing histological laboratory methods and equipment.
References
Full-Text: (1853 Views)
1. Introduction
Osteoarthritis (OA) is a chronic debilitating inflammation of joints characterized by destruction of articular cartilage which gradually becomes soft and frayed. Thickness of cartilage is reduced and subchondral bone eburnation and clustering of osteophytes will result in immobilization of joints and pain [1, 2]. Age, sex, genetic factors, nutrition, joint injuries and deformities and intense exercise are known risk factors for OA [3]. Many researchers believe that sport activities can damage the knee joint and may end in knee OA [4-6]. In this regard, physical activity for a long time may lead to the joint disease (as sport injury) [7] and intense exercises may be harmful [8, 9]. On the other hand, some researchers recommend on moderate intensity exercises [10-13]. However, low and medium exercises may also exert pressure on the joint cartilage. Some studies have shown that these forces inhibit interleukin (IL)-1β, anti-inflammatory signals, lipopolysaccharide-induced pro-inflammatory gene transcription, and Tumor Necrosis Factor (TNF)-α in in-vitro systems [14-16].
Some human studies have evaluated several treatment protocols by using the questionnaires or assessing some special physical activities. However because of human ethical issues, the intervention effects on various joint cartilage parameters such as its biochemical properties are unclear [17]. Therefore, by using some animal models such as mice or rats, it is possible to assess the histopathological changes in the joints with OA. It was shown that the pathogenesis and results of these studies can be generalized to the human diseases [18].
Histological studies are more accurate ways to detect the severity of OA at specific sites in humans and other species. For this purpose, histopathology analysis can be done on cartilage biopsy samples taken from joint of animals and humans in vivo. However, there are many limitations to collect whole tissue for standardization and grading of micro-molecular changes occurring in normal histological architecture. Experimental OA animal models are used for better understanding of disease progression and efficacy of treatments. Moreover, simple and reproducible histopathology grading of experimentally induced OA in animals provide necessary information about structural changes in joints as well as environmental or biological risk factors [19]. Thus, this study aimed to compare histopathological changes after 6 weeks moderate exercise-induced experimental OA model in male and female.
2. Materials and Methods
All steps of this study were approved by the Animal Ethics Committee, based on the Research Council of the Veterinary School, Shahid Bahonar University of Kerman, Iran.
Sixteen, 9 to 10-week-old male Wistar rats, and also sixteen 10 to 11-week-old, female Wistar rats, weighing 190 to 200 g, were randomly assigned to four equal groups (2 male and 2 female groups) and housed in metabolic cages with free access to commercial rodent diet and water. All rats were allowed a 5-day adaptation period in a room with controlled conditions (temperature 22°C–25°C and humidity of 60–70%) before starting the experiment. This protocol was done based on international guiding ethics for biomedical researchers involving laboratory animals, at the Veterinary Medicine Faculty, Shahid Bahonar University of Kerman, Iran.
Eight of each sex were selected as control groups and kept separately. Running exercises were performed in remained 16 male and female Wistar rats. One week after adapting period, both male and female groups commenced 6 weeks of exercise on a motor-driven treadmill (Model T510, DRI Co., Taoyuan, Taiwan). The speed was set at 18-20 m/min and the rats ran on the treadmill for 60 minutes each day, 5 days per week. In other words, the running load was set at 1000 m/day for a 5 day/week program. The treadmill incline was zero. This intensity was considered at a moderate level of exercise [20].
On day 43, the animals were killed by cervical dislocation under anesthesia. Their whole knee joints were decalcified with 10% acid formic. Then the samples were fixed in 10% neutral buffered formalin solution, dehydrated in graded anhydrous absolute ethanol and xylol, embedded in paraffin wax, and sectioned with 5 μm thickness. Sections were then stained using hematoxylin-eosin to observe the cellularity with light microscope. It is noticeable that frontal and sagittal sections were prepared from medial condyle of the femur.
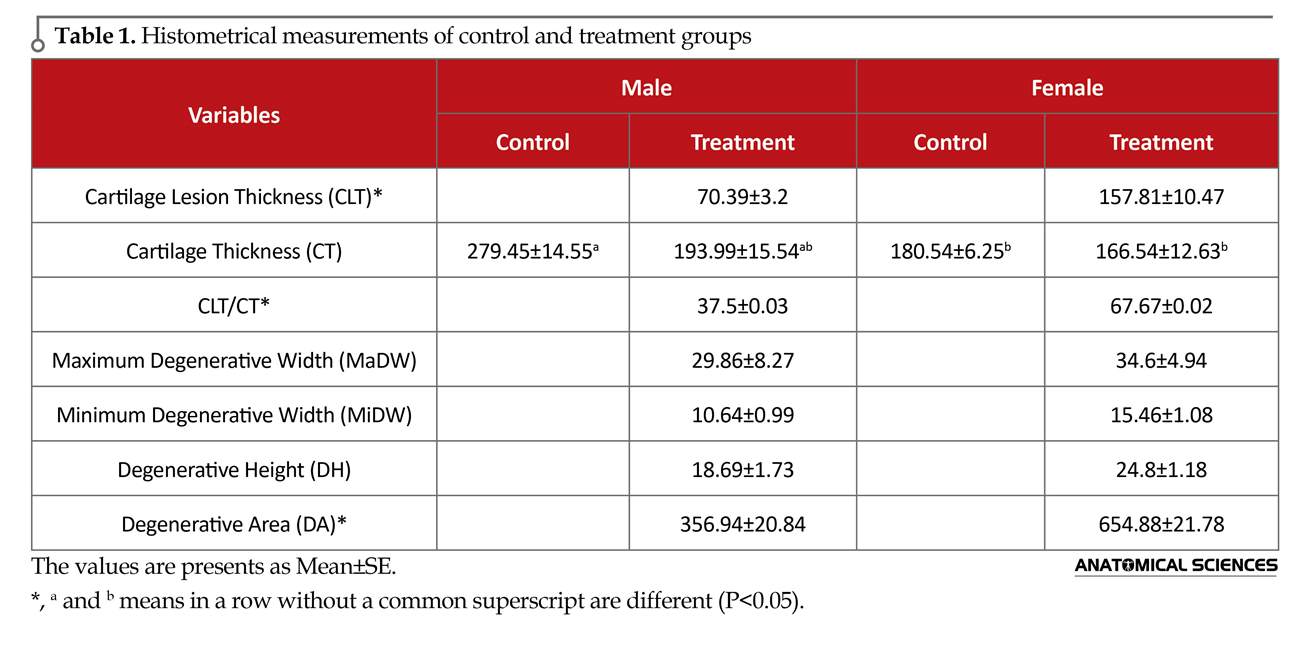
The severity of OA lesions was graded on a scale adopted from OARSI (Osteoarthritis Research Society International) histopathology instructions. Three histopathological measures, including cartilage lesion thickness (CLT: including areas of chondrocyte and proteoglycan loss with chondrocyte with pyknotic nucleus or absent nuclei), Cartilage Thickness (CT), Maximum Degeneration Width (MaDW), Minimum Degeneration Width (MiDW) and Degeneration Height (DH) were measured [7, 21, 22] by a digital system (Dino-eye, AM-7023, 5Mp, Taiwan) (Figure 1 and 2). Also the CLT/CT ratio was evaluated in experimental groups.
According to the shape of the degenerative areas (about a trapezoidal shape) (Figure 2), the area of each degenerative region was calculated via this formula: DA=0.5 (MaDW+MiDW) DH. Histomorphological scores in micrometers were assigned to these measurements for statistical analysis [23]. Results were expressed as Means±SE (Table 1). Data were analyzed by 1-way ANOVA followed by Tukey test for CT and the Independent t test for other parameters, using SPSS 16 (SPSS; Chicago, USA). P values less than 0.05 were considered statistically significant.
3. Results
In the control group, the cartilage was intact. The chondrocytes were placed in their lacunae. These cells presented a granular cytoplasm with discrete vacuoles that were surrounded by cartilaginous capsule and a nucleus in the central part of the cell (Figure 1). The knee joints of female rats with a total running load of 30 km with average running speed 18-20 m/h, showed marked histological changes, typically with fibrillations and clefts in joint cartilage, cell cloning, and moderate or severe reduction in the H&E staining (Figure 1).
In male rats with a total running load as the females, OA was less noticeable. However, the histological modifications with some superficial irregularities, cloning, and mild reduction in the H&E staining were visible. Comparison of the extent of histological changes within the two running groups of rats revealed significant differences with an increasing mild to moderate osteoarthritis in them compared to the control groups. On the other hand, histopathological changes in the knee of female rats were more noticeable than those in the male groups. The results of CLT, CT, CLT/CT, MaDW, MiDW, DH and DA are summarized and presented in Table 1.
4. Discussion
This study aimed to verify if sole, excessive running load leads to osteoarthritic changes in knee of female and male Wistar rats and evaluation a same moderate term treadmill exercise (a similar exercise program) on the extent of osteoarthritic changes in both equiponderant sexes.
Histological grading and grouping of the results into four classes (no, mild, moderate, and severe OA) [24] confirmed a significant increase in osteoarthritic changes in running female group while no changes were seen in the female controls. The histological findings of this group were categorized as mild OA. In the running male rats with a total running distance as the females’, most knee joints showed no OA. A noticeable result in our study was that both male and female treadmill exercise groups showed reduction in their total cartilage thickness compared to their matched controls (Table 1).
In our study, we could show by histological assessment that moderate running load leads to the development of OA in the knee joints of both female and male Wistar rats. Hereby, the grade of OA strongly depends on the extent of the joint load with increasing OA in females. Based on some studies, running load must be extremely high to cause even mild or moderate OA [21, 24, 25]. Our results showed that treadmill exercise for a speed set at 18-20 m/min for 60 minutes each day on 5 days of every week for 6 weeks leads to mild OA in female rats.
Osteoarthritis (OA) is a chronic debilitating inflammation of joints characterized by destruction of articular cartilage which gradually becomes soft and frayed. Thickness of cartilage is reduced and subchondral bone eburnation and clustering of osteophytes will result in immobilization of joints and pain [1, 2]. Age, sex, genetic factors, nutrition, joint injuries and deformities and intense exercise are known risk factors for OA [3]. Many researchers believe that sport activities can damage the knee joint and may end in knee OA [4-6]. In this regard, physical activity for a long time may lead to the joint disease (as sport injury) [7] and intense exercises may be harmful [8, 9]. On the other hand, some researchers recommend on moderate intensity exercises [10-13]. However, low and medium exercises may also exert pressure on the joint cartilage. Some studies have shown that these forces inhibit interleukin (IL)-1β, anti-inflammatory signals, lipopolysaccharide-induced pro-inflammatory gene transcription, and Tumor Necrosis Factor (TNF)-α in in-vitro systems [14-16].
Some human studies have evaluated several treatment protocols by using the questionnaires or assessing some special physical activities. However because of human ethical issues, the intervention effects on various joint cartilage parameters such as its biochemical properties are unclear [17]. Therefore, by using some animal models such as mice or rats, it is possible to assess the histopathological changes in the joints with OA. It was shown that the pathogenesis and results of these studies can be generalized to the human diseases [18].
Histological studies are more accurate ways to detect the severity of OA at specific sites in humans and other species. For this purpose, histopathology analysis can be done on cartilage biopsy samples taken from joint of animals and humans in vivo. However, there are many limitations to collect whole tissue for standardization and grading of micro-molecular changes occurring in normal histological architecture. Experimental OA animal models are used for better understanding of disease progression and efficacy of treatments. Moreover, simple and reproducible histopathology grading of experimentally induced OA in animals provide necessary information about structural changes in joints as well as environmental or biological risk factors [19]. Thus, this study aimed to compare histopathological changes after 6 weeks moderate exercise-induced experimental OA model in male and female.
2. Materials and Methods
All steps of this study were approved by the Animal Ethics Committee, based on the Research Council of the Veterinary School, Shahid Bahonar University of Kerman, Iran.
Sixteen, 9 to 10-week-old male Wistar rats, and also sixteen 10 to 11-week-old, female Wistar rats, weighing 190 to 200 g, were randomly assigned to four equal groups (2 male and 2 female groups) and housed in metabolic cages with free access to commercial rodent diet and water. All rats were allowed a 5-day adaptation period in a room with controlled conditions (temperature 22°C–25°C and humidity of 60–70%) before starting the experiment. This protocol was done based on international guiding ethics for biomedical researchers involving laboratory animals, at the Veterinary Medicine Faculty, Shahid Bahonar University of Kerman, Iran.
Eight of each sex were selected as control groups and kept separately. Running exercises were performed in remained 16 male and female Wistar rats. One week after adapting period, both male and female groups commenced 6 weeks of exercise on a motor-driven treadmill (Model T510, DRI Co., Taoyuan, Taiwan). The speed was set at 18-20 m/min and the rats ran on the treadmill for 60 minutes each day, 5 days per week. In other words, the running load was set at 1000 m/day for a 5 day/week program. The treadmill incline was zero. This intensity was considered at a moderate level of exercise [20].
On day 43, the animals were killed by cervical dislocation under anesthesia. Their whole knee joints were decalcified with 10% acid formic. Then the samples were fixed in 10% neutral buffered formalin solution, dehydrated in graded anhydrous absolute ethanol and xylol, embedded in paraffin wax, and sectioned with 5 μm thickness. Sections were then stained using hematoxylin-eosin to observe the cellularity with light microscope. It is noticeable that frontal and sagittal sections were prepared from medial condyle of the femur.
The severity of OA lesions was graded on a scale adopted from OARSI (Osteoarthritis Research Society International) histopathology instructions. Three histopathological measures, including cartilage lesion thickness (CLT: including areas of chondrocyte and proteoglycan loss with chondrocyte with pyknotic nucleus or absent nuclei), Cartilage Thickness (CT), Maximum Degeneration Width (MaDW), Minimum Degeneration Width (MiDW) and Degeneration Height (DH) were measured [7, 21, 22] by a digital system (Dino-eye, AM-7023, 5Mp, Taiwan) (Figure 1 and 2). Also the CLT/CT ratio was evaluated in experimental groups.
According to the shape of the degenerative areas (about a trapezoidal shape) (Figure 2), the area of each degenerative region was calculated via this formula: DA=0.5 (MaDW+MiDW) DH. Histomorphological scores in micrometers were assigned to these measurements for statistical analysis [23]. Results were expressed as Means±SE (Table 1). Data were analyzed by 1-way ANOVA followed by Tukey test for CT and the Independent t test for other parameters, using SPSS 16 (SPSS; Chicago, USA). P values less than 0.05 were considered statistically significant.
3. Results
In the control group, the cartilage was intact. The chondrocytes were placed in their lacunae. These cells presented a granular cytoplasm with discrete vacuoles that were surrounded by cartilaginous capsule and a nucleus in the central part of the cell (Figure 1). The knee joints of female rats with a total running load of 30 km with average running speed 18-20 m/h, showed marked histological changes, typically with fibrillations and clefts in joint cartilage, cell cloning, and moderate or severe reduction in the H&E staining (Figure 1).
In male rats with a total running load as the females, OA was less noticeable. However, the histological modifications with some superficial irregularities, cloning, and mild reduction in the H&E staining were visible. Comparison of the extent of histological changes within the two running groups of rats revealed significant differences with an increasing mild to moderate osteoarthritis in them compared to the control groups. On the other hand, histopathological changes in the knee of female rats were more noticeable than those in the male groups. The results of CLT, CT, CLT/CT, MaDW, MiDW, DH and DA are summarized and presented in Table 1.
4. Discussion
This study aimed to verify if sole, excessive running load leads to osteoarthritic changes in knee of female and male Wistar rats and evaluation a same moderate term treadmill exercise (a similar exercise program) on the extent of osteoarthritic changes in both equiponderant sexes.
Histological grading and grouping of the results into four classes (no, mild, moderate, and severe OA) [24] confirmed a significant increase in osteoarthritic changes in running female group while no changes were seen in the female controls. The histological findings of this group were categorized as mild OA. In the running male rats with a total running distance as the females’, most knee joints showed no OA. A noticeable result in our study was that both male and female treadmill exercise groups showed reduction in their total cartilage thickness compared to their matched controls (Table 1).
In our study, we could show by histological assessment that moderate running load leads to the development of OA in the knee joints of both female and male Wistar rats. Hereby, the grade of OA strongly depends on the extent of the joint load with increasing OA in females. Based on some studies, running load must be extremely high to cause even mild or moderate OA [21, 24, 25]. Our results showed that treadmill exercise for a speed set at 18-20 m/min for 60 minutes each day on 5 days of every week for 6 weeks leads to mild OA in female rats.
Our obtained results revealed that females show more cartilage changes in a moderate exercise compared to males with the same body weight. Some studies indicate that the knee cartilage volume differences are noticeable in adults aged between 26 and 61 years. Based on this study, radiographic assessments revealed that men’s cartilage volume is 33%–42% greater than that in women in normal cases [26]. Our study revealed that male rats had at least about 35% greater cartilage volume than females in the control groups. In contrast with our results, Faber et al. [27] reported that the volume of knee cartilage is more than that in the females. They believed that sex differences didn’t significantly affect the cartilage volume after matching body weight and height. Ding et al. reported that body and bone size have significant effect on this difference, which is in agreement with some ideas that cartilage volume has a direct relationship with bone size [28-30]. In other words, males have a thicker cartilage than females.
In our study, we showed some significant changes in the same body weight and age. However, physical activity measurements and fitness did not account for the detected differences. The differences that related to the sex declined but continued at all parts in the whole of the joint significantly. It suggests that other parameters may explain the sex differences. This may be because of limitations in bone measurements. However, these measures are highly reproducible, best reflect the surface and structure of the joint. Therefore, the growth factors and sex hormones are the most important candidates to explain this difference. Presence of the receptors of progesterone, estrogen and testosterone – some of the sex hormones – are reported in the human fetal cartilaginous tissue [31]. It has been shown that androgens secretion can stimulate the proliferation of chondrocytes. These cells produce and release the collagen and proteoglycans that are necessary to conserve and sustain the cartilage [32]. It has been shown that hormone therapy in post-menopausal women leads to increase in the cartilage volume [33].
Some research studies revealed that blood serum testosterone has a positive direct effect on the cartilage volume of the knee joint at all sites [34]. Also growth factors such as transforming growth factor-ɞ and insulin-like growth factor-1 have important effect in development of the articular cartilage and proteoglycan production [35]. It is noticeable that evaluating the effect of sex hormone and growth factors on the cartilage formation and its volume needs more investigation. Our results showed significant changes in the knee cartilage after a moderate exercise in male and female rats with similar age and weight. It also suggests that the exercise program in females should be different from that in males.
Ethical Considerations
Compliance with ethical guidelines
All steps of this study were approved by the Animal Ethics Committee, based on the Research Council of the Veterinary School, Shahid Bahonar University of Kerman, Iran.
Funding
This research was financially supported by the Research Council of Shahid Bahonar University of Kerman (No: 1.7.1395).
Conflict of interest
The authors declared no conflict of interest.
Acknowledgements
The authors would like to thank Mr. Mazhab Jaafari for providing histological laboratory methods and equipment.
References
- Jones G, Ding C, Scott F, Glisson M, Cicuttini F. Early radiographic osteoarthritis is associated with substantial changes in cartilage volume and tibial bone surface area in both males and females. Osteoarthritis and Cartilage. 2004; 12(2):169–74. [DOI:10.1016/j.joca.2003.08.010]
- Zhang W, Moskowitz RW. OARSI recommendations for the management of hip and knee osteoarthritis, Part II: OARSI evidence-based, expert consensus guidelines. Osteoarthritis and Cartilage. 2008; 16(2):137-62. [DOI:10.1016/j.joca.2007.12.013]
- Bronner F, Farach Carson M. Bone and osteoarthritis. Berlin: Springer; 2007. [DOI:10.1007/978-1-84628-701-5]
- Klug M, Shrier I, McBain K, Shultz R, Meeuwisse WH, Garza D, Matheson GO. The prevention of sport injury: An anlaysis of 12000 published manuscripts. Clinical Journal of Sport Medicine. 2010; 20(6):407–12. [DOI:10.1097/JSM.0b013e3181f4a99c]
- Mikkelsson O, Nupponen H, Kaprio J, Kautiainen H, Mikkelsson M, Kujala M. Adolescent flexibility, endurance strength, and physical activity as predictors of adult tension neck, low back pain, and knee injury: A 25 year follow up study. British Journal of Sports Medicine. 2006; 40(2):107-13. [DOI:10.1136/bjsm.2004.017350]
- Giza E, Mithöfer K, Farrell L, Zarins B, Gill T. Injuries in women’s professional soccer. British Journal of Sports Medicine. 2005; 39(4):212-6. [DOI:10.1136/bjsm.2004.011973]
- Fallah Mohammadi M, Hajizadeh Moghaddam A, Mirkarimpur H. The effects of a moderate exercise program on knee osteoarthritis in male Wistar rats. Iranian Journal of Basic Medical Science. 2013; 16(5):683-8. [PMCID] [PMID]
- Blagojevic M, Jinks C, Jeffery A, Jordan KP. Risk factors for onset of osteoarthritis of the knee in older adults: A systematic review and meta-analysis. Osteoarthritis and Cartilage. 2010; 18(1):24-33. [DOI:10.1016/j.joca.2009.08.010]
- Lee YJ, Park JA, Yang SH, Kim KY, Kim BK, Lee EY, et al. Evaluation of osteoarthritis induced by treadmill-running exercise using the modified Mankin and the new OARSI assessment system. Rheumatology International. 2011; 31(12):1571-6. [DOI:10.1007/s00296-010-1520-4]
- Cifuentes D, Rocha LG, Silva LA, Brito AC, Rueff-Barroso CR, Porto LC, et al. Decrease in oxidative stress and histological changes induced by physical exercise calibrated in rats with osteoarthritis induced by monosodium iodoacetate. Osteoarthritis and Cartilage. 2010; 18(8):1088-95. [DOI:10.1016/j.joca.2010.04.004]
- Vignon E, Valat JP, Rossignol M, Avouac B, Rozenberg S, Thoumie P, et al. Osteoarthritis of the review and synthesis (OASIS). Joint Bone Spine. 2006; 73(4):442–55. [DOI:10.1016/j.jbspin.2006.03.001]
- Sutton AJ, Muir KR, Mockett S, Fentem P. A case-control study to investigate the relation between low and moderate levels of physical activity and osteoarthritis of the knee using data collected as part of the Allied Dunbar National Fitness Survey. Annals Rheumatology Disease. 2001; 60(8):756-64. [DOI:10.1136/ard.60.8.756]
- Kazeem A, Olubayo A, Ganiyu A. Plasma Nitric Oxide and Acute Phase Proteins after moderate and prolonged exercises. Iranian Journal of Basic Medicine Science. 2012; 15(1):602-7. [PMCID] [PMID]
- Fehrenbacher A, Steck E, Rickert M, Roth W, Richter W. Rapid regulation of collagen but not metalloproteinase 1, 3, 13, 14 and tissue inhibitor of metalloproteinase 1, 2, 3 expression in response to mechanical loading of cartilage explants in vitro. Archives Biochemistry Biophysics. 2003; 410(1):39-47. [DOI:10.1016/S0003-9861(02)00658-6]
- Mio K, Saito S, Tomatsu T, Toyama Y. Intermittent compressive strain may reduce aggrecanase expression in cartilage: A study of chondrocytes in agarose gel. Clinical Orthopaedics and Related Research. 2005; 433:225-32. [DOI:10.1097/01.blo.0000150466.30696.c6]
- Park S, Hung CT, Ateshian GA. Mechanical response of bovine articular cartilage under dynamic unconfined compression loading at physiological stress levels. Osteoarthritis and Cartilage 2004; 12(1):65-73. [DOI:10.1016/j.joca.2003.08.005]
- Roos EM, Dahlberg L. Positive effects of moderate exercise on glycosaminoglycan content in knee cartilage: A four-month, randomized, controlled trial in patients at risk of osteoarthritis. Arthritis Rheumatism. 2005; 52(11):3507–14. [DOI:10.1002/art.21415]
- Bendele AM. Animal models of osteoarthritis. Journal of Musculoskeletal Neuronal Interactions. 2001; 1(4):363-76. [PMID]
- Khan HM, Ashraf M, Hashmi AS, Ahmad MUD, Anjum AA. Papain induced progressive degenerative changes in articular cartilage of rat femorotibial joint and its histopathological grading. The Journal of Animal and Plant Sciences. 2013; 23(2):350-8.
- Iwamoto J, Takeda T, Ichimura S: Effect of exercise on tibial and lumbar vertebral bone mass in mature osteopenic rats: Bone histomorphometry study. Journal of Orthopaedics Science. 1998; 3(5):257-63. [DOI:10.1007/s007760050051]
- Ramezani M, Alizadeh MH, Kordi MR. Effect of intensity and volume of endurance training on the incidence of knee osteoarthritis in healthy male rats. Razi Journal of Medical Sciences. 2015; 22(131):97-105.
- Gerwin N, Bendele AM, Glasson S, Carlson CS. The OARSI histopathology initiative – recommendations for histological assessments of osteoarthritis in the rat. Osteoarthritis and Cartilage. 2010; 18(3):S24–S34. [DOI:10.1016/j.joca.2010.05.030]
- Hadipur M, Mozaffari R. Protective effects of nutrients on articular cartilage of rats and its histological assessment. Journal of Daneshvar Pezeshki. 2008; 15:88-94.
- Lequesne MG, Dang N, Lane NE. Sport practice and osteoarthritis of the limbs. Osteoarthritis and Cartilage. 1997; 5(2):75-86. [DOI:10.1016/S1063-4584(97)80001-5]
- Pap G, Eberhardt R, Sturmer I, Machner A, Schwarzberg H, Roessner A, et al. Development of osteoarthritis in the knee joints of Wistar rats after strenuous running exercise in a running wheel by intracranial self-stimulation. Pathology Research Practice. 1998; 194(1):41-7. [DOI:10.1016/S0344-0338(98)80010-1]
- Ding C, Cicuttini F, Scott F, Glisson M, Jones G. Sex differences in knee cartilage volume in adults: role of body and bone size, age and physical activity. Rheumatology. 2003; 42(11):1317–23. [DOI:10.1093/rheumatology/keg374]
- Faber SC, Eckstein F, Lukasz S, Muhlbauer R, Hohe J, Englmeier KH, et al. Sex differences in knee joint cartilage thickness, volume and articular surface areas: Assessment with quantitative three-dimensional MR imaging. Skeletal Radiology. 2001; 30(3):144–50. [DOI:10.1007/s002560000320]
- Eckstein F, Winzheimer M, Hohe J, Englmeier KH, Reiser M. Interindividual variability and correlation among morphological parameters of knee joint cartilage plates: Analysis with three-dimensional MR imaging. Osteoarthritis and Cartilage. 2001; 9(2):101–11. [DOI:10.1053/joca.2000.0365]
- Jones G, Glisson M, Hynes K, Cicuttini F. Sex and site differences in cartilage development: A possible explanation for variations in knee osteoarthritis in later life. Arthritis Rheumatology. 2000; 43(11):2543–8. [DOI:10.1002/1529-0131(200011)43:113.0.CO;2-K]
- Ding CH, Cicuttini F, Scott F, Glisson M, Jones G. A cross-sectional study of the determinants of knee cartilage volume. Osteoarthritis Cartilage. 2002; 10(Suppl A):S57.
- Ben-Hur H, Thole HH, Mashiah A et al. Estrogen, progesterone and testosterone receptors in human fetal cartilaginous tissue: Immunohistochemical studies. Calcified Tissue International. 1997; 60(6):520–6. [DOI:10.1007/s002239900274]
- Franchimont P, Bassleer C. Effects of hormones and local growth factors on articular chondrocyte metabolism. Journal of Rheumatology. 1991; 27(Suppl.):68–70. [PMID]
- Wluka AE, Davis SR, Bailey M, Stuckey SL, Cicuttini FM. Users of oestrogen replacement therapy have more knee cartilage than non-users. Annals Rheumatology Disease. 2001; 60(4):332–6. [DOI:10.1136/ard.60.4.332]
- Cicuttini FM, Wluka A, Bailey M, Sullivan RO, Poon C, Yeung S, et al. Factors affecting knee cartilage volume in healthy men. Rheumatology. 2003; 42(2):258–62. [DOI:10.1093/rheumatology/keg073]
- van der Kraan PM, Buma P, van Kuppevelt T, van den Berg WB. Interaction of chondrocytes, extracellular matrix and growth factors: Relevance for articular cartilage tissue engineering. Osteoarthritis and Cartilage. 2002; 10(8):631–7. [DOI:10.1053/joca.2002.0806]
Type of Study: Original |
Subject:
Gross Anatomy
Received: 2017/11/10 | Accepted: 2018/04/25 | Published: 2018/07/1
Received: 2017/11/10 | Accepted: 2018/04/25 | Published: 2018/07/1
References
1. Jones G, Ding C, Scott F, Glisson M, Cicuttini F. Early radiographic osteoarthritis is associated with substantial changes in cartilage volume and tibial bone surface area in both males and females. Osteoarthritis and Cartilage. 2004; 12(2):169–74. [DOI:10.1016/j.joca.2003.08.010] [DOI:10.1016/j.joca.2003.08.010]
2. Zhang W, Moskowitz RW. OARSI recommendations for the management of hip and knee osteoarthritis, Part II: OARSI evidence-based, expert consensus guidelines. Osteoarthritis and Cartilage. 2008; 16(2):137-62. [DOI:10.1016/j.joca.2007.12.013] [DOI:10.1016/j.joca.2007.12.013]
3. Bronner F, Farach Carson M. Bone and osteoarthritis. Berlin: Springer; 2007. [DOI:10.1007/978-1-84628-701-5] [DOI:10.1007/978-1-84628-701-5]
4. Klug M, Shrier I, McBain K, Shultz R, Meeuwisse WH, Garza D, Matheson GO. The prevention of sport injury: An anlaysis of 12000 published manuscripts. Clinical Journal of Sport Medicine. 2010; 20(6):407–12. [DOI:10.1097/JSM.0b013e3181f4a99c] [DOI:10.1097/JSM.0b013e3181f4a99c]
5. Mikkelsson O, Nupponen H, Kaprio J, Kautiainen H, Mikkelsson M, Kujala M. Adolescent flexibility, endurance strength, and physical activity as predictors of adult tension neck, low back pain, and knee injury: A 25 year follow up study. British Journal of Sports Medicine. 2006; 40(2):107-13. [DOI:10.1136/bjsm.2004.017350] [DOI:10.1136/bjsm.2004.017350]
6. Giza E, Mithöfer K, Farrell L, Zarins B, Gill T. Injuries in women's professional soccer. British Journal of Sports Medicine. 2005; 39(4):212-6. [DOI:10.1136/bjsm.2004.011973] [DOI:10.1136/bjsm.2004.011973]
7. Fallah Mohammadi M, Hajizadeh Moghaddam A, Mirkarimpur H. The effects of a moderate exercise program on knee osteoarthritis in male Wistar rats. Iranian Journal of Basic Medical Science. 2013; 16(5):683-8. [PMCID] [PMID] [PMID] [PMCID]
8. Blagojevic M, Jinks C, Jeffery A, Jordan KP. Risk factors for onset of osteoarthritis of the knee in older adults: A systematic review and meta-analysis. Osteoarthritis and Cartilage. 2010; 18(1):24-33. [DOI:10.1016/j.joca.2009.08.010] [DOI:10.1016/j.joca.2009.08.010]
9. Lee YJ, Park JA, Yang SH, Kim KY, Kim BK, Lee EY, et al. Evaluation of osteoarthritis induced by treadmill-running exercise using the modified Mankin and the new OARSI assessment system. Rheumatology International. 2011; 31(12):1571-6. [DOI:10.1007/s00296-010-1520-4] [DOI:10.1007/s00296-010-1520-4]
10. Cifuentes D, Rocha LG, Silva LA, Brito AC, Rueff-Barroso CR, Porto LC, et al. Decrease in oxidative stress and histological changes induced by physical exercise calibrated in rats with osteoarthritis induced by monosodium iodoacetate. Osteoarthritis and Cartilage. 2010; 18(8):1088-95. [DOI:10.1016/j.joca.2010.04.004] [DOI:10.1016/j.joca.2010.04.004]
11. Vignon E, Valat JP, Rossignol M, Avouac B, Rozenberg S, Thoumie P, et al. Osteoarthritis of the review and synthesis (OASIS). Joint Bone Spine. 2006; 73(4):442–55. [DOI:10.1016/j.jbspin.2006.03.001] [DOI:10.1016/j.jbspin.2006.03.001]
12. Sutton AJ, Muir KR, Mockett S, Fentem P. A case-control study to investigate the relation between low and moderate levels of physical activity and osteoarthritis of the knee using data collected as part of the Allied Dunbar National Fitness Survey. Annals Rheumatology Disease. 2001; 60(8):756-64. [DOI:10.1136/ard.60.8.756] [DOI:10.1136/ard.60.8.756]
13. Kazeem A, Olubayo A, Ganiyu A. Plasma Nitric Oxide and Acute Phase Proteins after moderate and prolonged exercises. Iranian Journal of Basic Medicine Science. 2012; 15(1):602-7. [PMCID] [PMID] [PMID] [PMCID]
14. Fehrenbacher A, Steck E, Rickert M, Roth W, Richter W. Rapid regulation of collagen but not metalloproteinase 1, 3, 13, 14 and tissue inhibitor of metalloproteinase 1, 2, 3 expression in response to mechanical loading of cartilage explants in vitro. Archives Biochemistry Biophysics. 2003; 410(1):39-47. [DOI:10.1016/S0003-9861(02)00658-6] [DOI:10.1016/S0003-9861(02)00658-6]
15. Mio K, Saito S, Tomatsu T, Toyama Y. Intermittent compressive strain may reduce aggrecanase expression in cartilage: A study of chondrocytes in agarose gel. Clinical Orthopaedics and Related Research. 2005; 433:225-32. [DOI:10.1097/01.blo.0000150466.30696.c6] [DOI:10.1097/01.blo.0000150466.30696.c6]
16. Park S, Hung CT, Ateshian GA. Mechanical response of bovine articular cartilage under dynamic unconfined compression loading at physiological stress levels. Osteoarthritis and Cartilage 2004; 12(1):65-73. [DOI:10.1016/j.joca.2003.08.005] [DOI:10.1016/j.joca.2003.08.005]
17. Roos EM, Dahlberg L. Positive effects of moderate exercise on glycosaminoglycan content in knee cartilage: A four-month, randomized, controlled trial in patients at risk of osteoarthritis. Arthritis Rheumatism. 2005; 52(11):3507–14. [DOI:10.1002/art.21415] [DOI:10.1002/art.21415]
18. Bendele AM. Animal models of osteoarthritis. Journal of Musculoskeletal Neuronal Interactions. 2001; 1(4):363-76. [PMID] [PMID]
19. Khan HM, Ashraf M, Hashmi AS, Ahmad MUD, Anjum AA. Papain induced progressive degenerative changes in articular cartilage of rat femorotibial joint and its histopathological grading. The Journal of Animal and Plant Sciences. 2013; 23(2):350-8.
20. Iwamoto J, Takeda T, Ichimura S: Effect of exercise on tibial and lumbar vertebral bone mass in mature osteopenic rats: Bone histomorphometry study. Journal of Orthopaedics Science. 1998; 3(5):257-63. [DOI:10.1007/s007760050051] [DOI:10.1007/s007760050051]
21. Ramezani M, Alizadeh MH, Kordi MR. Effect of intensity and volume of endurance training on the incidence of knee osteoarthritis in healthy male rats. Razi Journal of Medical Sciences. 2015; 22(131):97-105.
22. Gerwin N, Bendele AM, Glasson S, Carlson CS. The OARSI histopathology initiative – recommendations for histological assessments of osteoarthritis in the rat. Osteoarthritis and Cartilage. 2010; 18(3):S24–S34. [DOI:10.1016/j.joca.2010.05.030] [DOI:10.1016/j.joca.2010.05.030]
23. Hadipur M, Mozaffari R. Protective effects of nutrients on articular cartilage of rats and its histological assessment. Journal of Daneshvar Pezeshki. 2008; 15:88-94.
24. Lequesne MG, Dang N, Lane NE. Sport practice and osteoarthritis of the limbs. Osteoarthritis and Cartilage. 1997; 5(2):75-86. [DOI:10.1016/S1063-4584(97)80001-5] [DOI:10.1016/S1063-4584(97)80001-5]
25. Pap G, Eberhardt R, Sturmer I, Machner A, Schwarzberg H, Roessner A, et al. Development of osteoarthritis in the knee joints of Wistar rats after strenuous running exercise in a running wheel by intracranial self-stimulation. Pathology Research Practice. 1998; 194(1):41-7. [DOI:10.1016/S0344-0338(98)80010-1] [DOI:10.1016/S0344-0338(98)80010-1]
26. Ding C, Cicuttini F, Scott F, Glisson M, Jones G. Sex differences in knee cartilage volume in adults: role of body and bone size, age and physical activity. Rheumatology. 2003; 42(11):1317–23. [DOI:10.1093/rheumatology/keg374] [DOI:10.1093/rheumatology/keg374]
27. Faber SC, Eckstein F, Lukasz S, Muhlbauer R, Hohe J, Englmeier KH, et al. Sex differences in knee joint cartilage thickness, volume and articular surface areas: Assessment with quantitative three-dimensional MR imaging. Skeletal Radiology. 2001; 30(3):144–50. [DOI:10.1007/s002560000320] [DOI:10.1007/s002560000320]
28. Eckstein F, Winzheimer M, Hohe J, Englmeier KH, Reiser M. Interindividual variability and correlation among morphological parameters of knee joint cartilage plates: Analysis with three-dimensional MR imaging. Osteoarthritis and Cartilage. 2001; 9(2):101–11. [DOI:10.1053/joca.2000.0365] [DOI:10.1053/joca.2000.0365]
29. Jones G, Glisson M, Hynes K, Cicuttini F. Sex and site differences in cartilage development: A possible explanation for variations in knee osteoarthritis in later life. Arthritis Rheumatology. 2000; 43(11):2543–8. [DOI:10.1002/1529-0131(200011)43:113.0.CO;2-K]
https://doi.org/10.1002/1529-0131(200011)43:11<2543::AID-ANR23>3.0.CO;2-K [DOI:10.1002/1529-0131(200011)43:113.0.CO;2-K]
30. Ding CH, Cicuttini F, Scott F, Glisson M, Jones G. A cross-sectional study of the determinants of knee cartilage volume. Osteoarthritis Cartilage. 2002; 10(Suppl A):S57.
31. Ben-Hur H, Thole HH, Mashiah A et al. Estrogen, progesterone and testosterone receptors in human fetal cartilaginous tissue: Immunohistochemical studies. Calcified Tissue International. 1997; 60(6):520–6. [DOI:10.1007/s002239900274] [DOI:10.1007/s002239900274]
32. Franchimont P, Bassleer C. Effects of hormones and local growth factors on articular chondrocyte metabolism. Journal of Rheumatology. 1991; 27(Suppl.):68–70. [PMID]
33. Wluka AE, Davis SR, Bailey M, Stuckey SL, Cicuttini FM. Users of oestrogen replacement therapy have more knee cartilage than non-users. Annals Rheumatology Disease. 2001; 60(4):332–6. [DOI:10.1136/ard.60.4.332] [DOI:10.1136/ard.60.4.332]
34. Cicuttini FM, Wluka A, Bailey M, Sullivan RO, Poon C, Yeung S, et al. Factors affecting knee cartilage volume in healthy men. Rheumatology. 2003; 42(2):258–62. [DOI:10.1093/rheumatology/keg073] [DOI:10.1093/rheumatology/keg073]
35. van der Kraan PM, Buma P, van Kuppevelt T, van den Berg WB. Interaction of chondrocytes, extracellular matrix and growth factors: Relevance for articular cartilage tissue engineering. Osteoarthritis and Cartilage. 2002; 10(8):631–7. [DOI:10.1053/joca.2002.0806] [DOI:10.1053/joca.2002.0806]
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |