Thu, Apr 25, 2024
Volume 17, Issue 1 (Winter & Spring 2020)
ASJ 2020, 17(1): 7-12 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Ahmadifar M, Panahian M, Vahidi Eyrisofla N. The Effects of Plant Extracts Mixture on Sperm Parameters and Damaged Testicular Tissue With Carbon Tetrachloride in Mice. ASJ 2020; 17 (1) :7-12
URL: http://anatomyjournal.ir/article-1-193-en.html
URL: http://anatomyjournal.ir/article-1-193-en.html
1- Department of Embryology, Royan Institute’s Reproductive Biomedicine, Royan Institute, Tehran, Iran.; Department of Biology, Faculty of Sciences, University of Science and Culture, Tehran, Iran.
2- Traditional Medicine Researcher, The University of Qom, Qom, Iran.
2- Traditional Medicine Researcher, The University of Qom, Qom, Iran.
Full-Text [PDF 586 kb]
(1393 Downloads)
| Abstract (HTML) (2817 Views)
Full-Text: (2972 Views)
1. Introduction
Approximately 10%-15% of couples are experiencing infertility. Although the use of health services for their treatment has increased in recent decades, the prevalence of infertility remained steady [1]. Moreover, infertility is a significant problem rising in families and communities. This issue is directly linked to the continuity of generations, and it has long been one of human wishes to raising their children. Thus, various treatment methods have been devised to solve this problem, depending on the cause of infertility that might include male or female or both. The number of infertile couples with azoospermia due in case of normal spermatogenesis and a good quality, open biopsy method Testicular sperm Extraction, Percutaneous Epididymis sperm Aspiration (TESE.PESA) or aspirate Fine Needle Aspiration (FNA) could be used where sperms from testis or epididymis are obtained, and after washing, injected to ovary. However, there is always the possibility of damage to sperm cells in the lack of special defense mechanism against oxidative agents [2].
Many studies on humans and laboratory animals have reported that substances, such as hypoxia, the alcoholic extract of Riccia fluitans, zinc deficiency, the use of drug colchicine or its injection into the epididymis, phoxim, diltiazem and methylene blue, ifosfamide of selenium deficiency in the diet cypermethrin brown of nicotine, cotinine and marijuana, alcohol, caffeine, and Capparis spinosa leaf extract reduce sperm motility [3-20]. Many studies used various herbal extracts on hormonal axis and testicular tissue. Extracts, such as rosemary, the alcoholic extracts of Centella asiatica, the alcoholic extract of fennel seeds, yarrow of aqueous extract of aerial parts Mirage, and aqueous extract of fenugreek seeds, decrease testosterone level and sometimes decrease LH level; thus, they could reduce the secondary sex characteristics, and majority of them decrease sperm density and reduce fertility. In addition, saffron extract, carrot seed extract, medicinal plants extract, garlic extract, marjoram, ginger, and velvet bean increase testosterone, LH, and sometimes FSH levels [21-33].
2. Materials and methods
In this study, 20 male Naval Medical Research Institute (NMRI) mice were used. They were grouped into the control and experimental groups. The control group received distilled water, and the treatment group was fed with 250 mg/kg of mixture of plant extract (Stipa capensis, Crocus sativus, pollen palm tree) after disruption with Carbon Tetrachloride (CCl4) for 60 days. The sample weight was measured at the onset and end of the experiment. In the treatment period, by anesthesia with ether, about 3 to 4 mL of blood from each mouse was collected from the left ventricle. The collected samples were centrifuged at 3000 rpm for 15 minutes. Then, using the sampler, isolated serums from whole blood were kept at -20°C for measuring the serum concentrations of LH, FSH, and testosterone. Hormonal assays were performed using the hormonal kits.
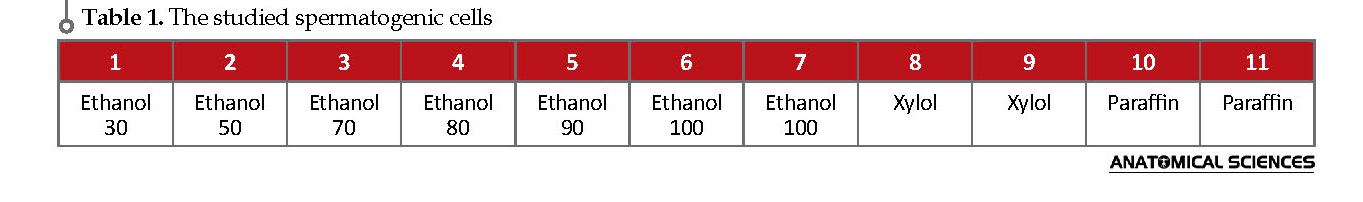
The testicles were removed and weight after the separation of surrounding connective tissues. Testicular tissue samples were stabilized in 10% buffered formalin solution. Then, testicles washed with normal saline into Catchers were maintained for 24 hours. Next, the specimens were fixed in the basket. For devices, jars with alcohol and paraffin and xylol were filled with a given program catchers devices for 1 hour each and were automatically applied. After dehydration, the samples were maintained in alcohol and xylol for one hour, twice. Then, the samples were stored in paraffin bath for 1 hour twice and got paraffin influenced (Table 1).
Paraffin molds were filled with paraffin melt, and the samples were placed in molds (horizontal or vertical). Paraffin samples were cooled and molded and labeled. Then, paraffin samples were removed from the baskets and kept in the freezer until sectioning. The paraffin samples took place in a special place on the microtome. After adjustment the screw of microtome, the film of paraffin, containing tissue was cut in 4 micron thickness. Then, the slices were lifted with two crossed brushes and placed in water bath (40-45°C) until the loss of paraffin. We transferred the samples in the slides and left them to dry. For staining the testis tissues, hematoxylin and eosin methods were used. Stained tissue sections were studied by optical microscopy camera. The studied spermatogenic cells (spermatogonia, primary spermatocytes, spermatids, spermatozoa, Sertoli) of different views X4, X10, X40, and X100) were used and the number of various experimental and control cells were compared.
3. Results
The effects of the mixture of plants in mice body and testis weights: The extract combined therapy increased the animal’s body and their testis weights in the treatment group, compared to the control group. The effects of the mixture of plants in sperm count: The count of sperm significantly increased in the treatment group compared with the controls. The effects of the mixture of plants in hormone levels: The level of LH had no significant differences between the control and treatment groups; however, the FSH and testosterone levels significantly increased in the treatment group (Figure 2). The effects of the mixture of plants in testis tissue: The histology analysis results suggested that both groups were normal in terms of appearance, shape, and distribution of seminiferous tubules (Figure 1).
Approximately 10%-15% of couples are experiencing infertility. Although the use of health services for their treatment has increased in recent decades, the prevalence of infertility remained steady [1]. Moreover, infertility is a significant problem rising in families and communities. This issue is directly linked to the continuity of generations, and it has long been one of human wishes to raising their children. Thus, various treatment methods have been devised to solve this problem, depending on the cause of infertility that might include male or female or both. The number of infertile couples with azoospermia due in case of normal spermatogenesis and a good quality, open biopsy method Testicular sperm Extraction, Percutaneous Epididymis sperm Aspiration (TESE.PESA) or aspirate Fine Needle Aspiration (FNA) could be used where sperms from testis or epididymis are obtained, and after washing, injected to ovary. However, there is always the possibility of damage to sperm cells in the lack of special defense mechanism against oxidative agents [2].
Many studies on humans and laboratory animals have reported that substances, such as hypoxia, the alcoholic extract of Riccia fluitans, zinc deficiency, the use of drug colchicine or its injection into the epididymis, phoxim, diltiazem and methylene blue, ifosfamide of selenium deficiency in the diet cypermethrin brown of nicotine, cotinine and marijuana, alcohol, caffeine, and Capparis spinosa leaf extract reduce sperm motility [3-20]. Many studies used various herbal extracts on hormonal axis and testicular tissue. Extracts, such as rosemary, the alcoholic extracts of Centella asiatica, the alcoholic extract of fennel seeds, yarrow of aqueous extract of aerial parts Mirage, and aqueous extract of fenugreek seeds, decrease testosterone level and sometimes decrease LH level; thus, they could reduce the secondary sex characteristics, and majority of them decrease sperm density and reduce fertility. In addition, saffron extract, carrot seed extract, medicinal plants extract, garlic extract, marjoram, ginger, and velvet bean increase testosterone, LH, and sometimes FSH levels [21-33].
2. Materials and methods
In this study, 20 male Naval Medical Research Institute (NMRI) mice were used. They were grouped into the control and experimental groups. The control group received distilled water, and the treatment group was fed with 250 mg/kg of mixture of plant extract (Stipa capensis, Crocus sativus, pollen palm tree) after disruption with Carbon Tetrachloride (CCl4) for 60 days. The sample weight was measured at the onset and end of the experiment. In the treatment period, by anesthesia with ether, about 3 to 4 mL of blood from each mouse was collected from the left ventricle. The collected samples were centrifuged at 3000 rpm for 15 minutes. Then, using the sampler, isolated serums from whole blood were kept at -20°C for measuring the serum concentrations of LH, FSH, and testosterone. Hormonal assays were performed using the hormonal kits.
The testicles were removed and weight after the separation of surrounding connective tissues. Testicular tissue samples were stabilized in 10% buffered formalin solution. Then, testicles washed with normal saline into Catchers were maintained for 24 hours. Next, the specimens were fixed in the basket. For devices, jars with alcohol and paraffin and xylol were filled with a given program catchers devices for 1 hour each and were automatically applied. After dehydration, the samples were maintained in alcohol and xylol for one hour, twice. Then, the samples were stored in paraffin bath for 1 hour twice and got paraffin influenced (Table 1).
Paraffin molds were filled with paraffin melt, and the samples were placed in molds (horizontal or vertical). Paraffin samples were cooled and molded and labeled. Then, paraffin samples were removed from the baskets and kept in the freezer until sectioning. The paraffin samples took place in a special place on the microtome. After adjustment the screw of microtome, the film of paraffin, containing tissue was cut in 4 micron thickness. Then, the slices were lifted with two crossed brushes and placed in water bath (40-45°C) until the loss of paraffin. We transferred the samples in the slides and left them to dry. For staining the testis tissues, hematoxylin and eosin methods were used. Stained tissue sections were studied by optical microscopy camera. The studied spermatogenic cells (spermatogonia, primary spermatocytes, spermatids, spermatozoa, Sertoli) of different views X4, X10, X40, and X100) were used and the number of various experimental and control cells were compared.
3. Results
The effects of the mixture of plants in mice body and testis weights: The extract combined therapy increased the animal’s body and their testis weights in the treatment group, compared to the control group. The effects of the mixture of plants in sperm count: The count of sperm significantly increased in the treatment group compared with the controls. The effects of the mixture of plants in hormone levels: The level of LH had no significant differences between the control and treatment groups; however, the FSH and testosterone levels significantly increased in the treatment group (Figure 2). The effects of the mixture of plants in testis tissue: The histology analysis results suggested that both groups were normal in terms of appearance, shape, and distribution of seminiferous tubules (Figure 1).
However, the average number of spermatogonia, primary spermatocytes, spermatids, and sperm increased significantly in the experimental group.
4. Discussion
The present study investigated the effect of mixed plant extracts on the hormonal axis and testes of adult male mice. Mixed plants, despite having many different compounds, can have numerous effects. The study results indicated that the mixed extract usage could lead to changes in testicular and pituitary-gonadal function and structure in mice. Bodyweight and testes were affected by testosterone [28, 32]. Previous studies have reported that testosterone directly affects Sertoli cells, liquid discharge tube, and several proteins, including growth factors and transferrin.
4. Discussion
The present study investigated the effect of mixed plant extracts on the hormonal axis and testes of adult male mice. Mixed plants, despite having many different compounds, can have numerous effects. The study results indicated that the mixed extract usage could lead to changes in testicular and pituitary-gonadal function and structure in mice. Bodyweight and testes were affected by testosterone [28, 32]. Previous studies have reported that testosterone directly affects Sertoli cells, liquid discharge tube, and several proteins, including growth factors and transferrin.
Moreover, a special role in sex cells was dividing power sexual cell division, and finally, their sperm production [34]. Furthermore, tubular atrophy and reduced sperm are signs of impaired spermatogenesis [35]. Many medicinal plants used in concentration can have an essential role in its effects. According to nurser, rosemary, at low doses, has no significant influence on spermatogenesis [21, 36]. We used the extracts of mixed herbs in this research. However, different combinations require accurate determination of regulatory pathways as well as combinations that could increase some hormones and sperm. Finally, applying the mixed extract dose was effective in the improvement of spermatogenesis. However, more investigations are required to identify the active ingredient and mechanism of action and efficacy of different doses of the extract.
The plant extracts had androgenic properties and increased androgen-dependent parameters. Moreover, it caused polyspermy and increased the weight of reproductive organs. The obtained results and further investigations could be beneficial for infertile patients and prevent or reduce the symptoms of menopause in men.
Ethical Considerations
Compliance with ethical guidelines
All ethical principles were considered in this article.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors contributions
Initial idea and costs: Mehdi panahian; Responsible for the tests: Mehdi Ahmadifar; Data analysis: Nazila vahidi.
Conflict of interest
The authors declared no conflict of interest.
Acknowledgments
Many thanks to the Royan Research Institute for collaborating.
References
Yao MWM, Schus DJ. Infertility. In: Berek JS. Novak's Gynecology. 13th ed. Philadelphia: Lippincott Williams & Wilkins; 2002.
Ball BA, Medina V, Gravance CG, Baumbe J. Effect of antioxidants on preservation of motility, viability and acrosomal integrity of equine spermatozoa during storage at 5 degrees C. Theriogenology. 2001; 56(4):577-89. [DOI:10.1016/S0093-691X(01)00590-8]
Kaur P, Bansal MP. Effect of experimental oxidative stress on steroidogenesis and DNA damage in mouse testis. Journal of Biomedical Science. 2004; 11(3):391-7. [DOI:10.1007/BF02254444] [PMID]
Sandhyakumary K, Bobby RG, Indira M. Antifertility effect of ricinus communis (Linn) on rats. Phytotherapy Research. 2003; 17(5):508-11. [DOI:10.1002/ptr.1308] [PMID]
Wei Q, Fan R, Yang X, Chen T. [Effect of zinc on reproductive toxicity in rats (Chinese)]. Wei Sheng Yan Jiu. 2003; 32(6):618-9. [PMID]
Ben-Chtrit A, Ben-Chtrit E, Nitzan R, Ron M. Colchicine inhibits spermatozoal motility in vitro. International journal of fertility and menopausal studies. 1993; 38(5):301-4. [PMID]
Schmid TE, Xu W, Adler ID. Detection of aneuploidy by multicolor FISH in mouse sperm after invivo treatment with acrylamide, colchicines, diazepam or thiabendazole. Mutagenesis. 1999; 14(2):173-9. [DOI:10.1093/mutage/14.2.173] [PMID]
Zhan N, Wang S, Wang X. [Effects of phoxim on sperm production and motility of rats (Chinese)]. Wei Shang Yan Jiu. 2000; 29(1):4-6. [PMID]
Wood BL, Doncel GF, Reddy PR, Sokal DC. Effect of diltiazem and methylen blue on human sperm motility, viability and cervical mucus penetration. Contraception. 2003; 67(3):241-5. [DOI:10.1016/S0010-7824(02)00515-2]
Ypsilantis P, Papaioannou N, Psalla D, Politou M, Simopoulos C. Effects of single dose administration of ifosfamide on tests and semen characteristics in the rabbit. Reproductive Toxicology. 2003; 17(2):237-45. [DOI:10.1016/S0890-6238(02)00127-2]
Ypsilantis P, Papaioannou N, Psalla D, Politou M, Pitiakoudis M, Simopoulos C. Effects of subchronic ifosfamide-mesna treatment on testes and semen characteristics in the rabbit. Reproductive Toxicology. 2003; 17(6):699-708. [DOI:10.1016/j.reprotox.2003.08.003]
Xu DX, Shen HM, Zhu QX, Chua L, Wang QN, Chia SE, et al. The associations among semen quality, oxidative DNA damage in human spermatozoa and concentrations of cadmium, lead and selenium in seminal plasma. Mutation Research/Genetic Toxicology and Environmental Mutagenesis. 2003; 534(1-2):155-63. [DOI:10.1016/S1383-5718(02)00274-7]
Elbetieha A, Da’as SI, Khamas W, Darmani H. Evaluation of the toxic potentials of cypermethrin pesticide on some reproductive and fertility parameters in the male rats. Archives of Environmental Contamination and Toxicology. 2001; 41(4):522-8. [DOI:10.1007/s002440010280] [PMID]
Adelusi B, al-Twaijiri MH, al-Meshari A, Kangave D, al-Nuaim LA, Younnus B. Correlation of smoking and coffee drinking with sperm progressive motility in infertile males. The African journal of medical sciences. 1988; 27(1-2):47-50. [PMID]
Mita P, Piatti E, Romano A, Magro B. [Epidemiology of male infertility (Italian)]. Archivio Italiano di Urologia e Andrologia. 1998; 70(2):85-91. [PMID]
Wang WY, Thomas CMG, Merkus HMWM, Zielhuis GA, Doesburg WH, Steegers-Theunissen RPM. Cigarette smoking and the risk of male factor subfertility: minor association between cotinine in seminal plasma and semen morphology. Fertility and Sterility. 2000; 74(5):930-5. [DOI:10.1016/S0015-0282(00)01567-3]
Marinelli D, Gaspari L, Pedotti P, Taioli E. Mini-review of studies on the effect of smoking and drinking habits on semen parameters. International Journal of Hygiene and Environmental Health. 2004; 207(3):185-92. [DOI:10.1078/1438-4639-00283] [PMID]
Weathersbee PS, Lodge JR. Caffeine: its direct and indirect influence on reproduction. The Journal of Reproductive Medicine. 1977; 19(2):55-63. [PMID]
Klonoff-Cohen H, Bleha J, Lam-Kruglick P. A prospective study of the effects of female and male caffeine consumption on the reproductive endpoints of IVF and gamete intra-fallopian transfer. Human Reproduction. 2002; 17(7):1746-54. [DOI:10.1093/humrep/17.7.1746] [PMID]
Aritajat S, Kaweewat K, Manosroi J, Manosroi A. Dominant lethal test in rats treated with some plant extracts. The Southeast Asian Journal of Tropical Medicine and Public Health. 2000;31(Suppl. 1):171-3. [PMID]
Heidari M, Vaezi GH, Ameri F, Ghaffari-Novin M, Keramati K, Rajaei F. [Antiandrogenic effects of Rosmarinus officinalis extract on the reproductive tract of male rats (Persian)]. Tehran Univerasity Medical Journal. 2008; 65(Suppl. 3):26-32.
Jasemi M, Saki Gh, Rahim F. [The effect of Centella asiataica alcoholic extract on the serum levels of testesterone, FSH and LH in male wistar rat (Persian)]. Journal of Sabzevar university of medical sciences. 2009; 16(1):6-11.
Salamatmanesh M, Shiravi A, Heydari nasrabady M. [The effect of (Anethum graveolens) seed alcoholic extract on spermatogenesis in male wistar ratsccc. Journal of animal biolaogy. 2009; 1(2):23-30.
Kerishchi P, Kazem P, Rouhani A, Roostaeean A. [Effect of Achillea millefolium L. extract on spermatogenesis and HaG axis in adult BALB/C mice (Persian)]. Yafte. 2004; 6(22):13a8.
Ahmadi A, Nasiri Nejad F, Parivar K. [Effect of aqueaous axtract of the aerial part of the ruta graveolens on the spermatogenesis of immature Balb/C mice (Persian)]. Razi Journal of Medical Sciences. 2007; 14(56):13-20.
Mokhtari M, Shariati M, Gharamani R. [Effect of Trigonella foenum-graecum L. seed extract on concentration of tesatesterone and spermatogenesis in rats (Persian)]. Journal of Medicinal Plants. 2008; 7(1):12-20.
Modaresi M, Messripoor M, Asadi Morghmaleki M, Hamedanian MK. [The effect of Saffron extract on testis tissue (Persian)]. Iranian Journal of Medicinal and Aromatic Plants. 2008; 24(2):237-43.
Nouri M, Khaki A, Fathi Azar F, Rashidi MR. The protective effects of carrot seed extract on spermatogenesis and cauda epididymal sperm reserves in gentamicin treated rats. Yakhteh Medical Journal. 2009; 11(3):327-32.
Naseri M, Heydari nasrabadi M, Khodarahmi P, Ahmadi F, Mojibi P, Abotalebei H. [Study of the effect of Fumaria parviflora alcoholic extract on spermatogenesis in male rats (Persian)]. New Cellular and Molecular Biotechnology Journal. 2011; 1(2):61-5.
Mirfard M, Johari H, Mokhtari M, Hematkhah V, Jamali H, Allahverdi Gh. [The effect of hydro-alcoholic garlic extract on testis weight and spermatogenesis in mature male rats under chemotherapy with cyclophosphamide (Persian)]. Journal of Fasa University of Medical Sciences. 2011; 1(3):123-30.
Kazemi P, Jowhary H, Sharifi E, Zeraatpishe A. [Androgenic effect of Origanum vulgarel L. spp viride extract on hormone level of pituitary-gonadal axis in mature male vistar rats (Persian)]. Journal of Arak University of Medical Sciences. 2012; 14(6):89-96.
Hemayatkhah Jahromi V, Parivar K, Forozanfar M. The effect of cinnamon extract on spermatogenesis hormonal axis of pituitary gonad in mice. Iranian Journal of Applied Animal Science. 2011; 1(2):99-103.
Chauhan NS, Saraf DK, Dixita VK. Effect of vajikaran rasayana herbs on pituitary-gonadal axis. European Journal of Integrative Medicine. 2010; 2(2):89-91. [DOI:10.1016/j.eujim.2010.03.002]
Carlson BM. Human embryology and developmental biology. 3rd ed. Philadelphia: Mosby; 2004.
Cai L, Hales BF, Robaire B. Induction of apoptosis in the germ cells of adult male rats after exposure to cyclophosphamide. Biology of Reproduction. 1997; 56(6):1490-7. [DOI:10.1095/biolreprod56.6.1490] [PMID]
Nusier MK, Bataineh HN, Daradkah HM. Adverse effects of rosemary (Rosmarinus officinalis L.) on reproductive function in adult male rats. Experimental Biology and Medicine (Maywood). 2007; 232(6):809-13. [PMID]
The plant extracts had androgenic properties and increased androgen-dependent parameters. Moreover, it caused polyspermy and increased the weight of reproductive organs. The obtained results and further investigations could be beneficial for infertile patients and prevent or reduce the symptoms of menopause in men.
Ethical Considerations
Compliance with ethical guidelines
All ethical principles were considered in this article.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors contributions
Initial idea and costs: Mehdi panahian; Responsible for the tests: Mehdi Ahmadifar; Data analysis: Nazila vahidi.
Conflict of interest
The authors declared no conflict of interest.
Acknowledgments
Many thanks to the Royan Research Institute for collaborating.
References
Yao MWM, Schus DJ. Infertility. In: Berek JS. Novak's Gynecology. 13th ed. Philadelphia: Lippincott Williams & Wilkins; 2002.
Ball BA, Medina V, Gravance CG, Baumbe J. Effect of antioxidants on preservation of motility, viability and acrosomal integrity of equine spermatozoa during storage at 5 degrees C. Theriogenology. 2001; 56(4):577-89. [DOI:10.1016/S0093-691X(01)00590-8]
Kaur P, Bansal MP. Effect of experimental oxidative stress on steroidogenesis and DNA damage in mouse testis. Journal of Biomedical Science. 2004; 11(3):391-7. [DOI:10.1007/BF02254444] [PMID]
Sandhyakumary K, Bobby RG, Indira M. Antifertility effect of ricinus communis (Linn) on rats. Phytotherapy Research. 2003; 17(5):508-11. [DOI:10.1002/ptr.1308] [PMID]
Wei Q, Fan R, Yang X, Chen T. [Effect of zinc on reproductive toxicity in rats (Chinese)]. Wei Sheng Yan Jiu. 2003; 32(6):618-9. [PMID]
Ben-Chtrit A, Ben-Chtrit E, Nitzan R, Ron M. Colchicine inhibits spermatozoal motility in vitro. International journal of fertility and menopausal studies. 1993; 38(5):301-4. [PMID]
Schmid TE, Xu W, Adler ID. Detection of aneuploidy by multicolor FISH in mouse sperm after invivo treatment with acrylamide, colchicines, diazepam or thiabendazole. Mutagenesis. 1999; 14(2):173-9. [DOI:10.1093/mutage/14.2.173] [PMID]
Zhan N, Wang S, Wang X. [Effects of phoxim on sperm production and motility of rats (Chinese)]. Wei Shang Yan Jiu. 2000; 29(1):4-6. [PMID]
Wood BL, Doncel GF, Reddy PR, Sokal DC. Effect of diltiazem and methylen blue on human sperm motility, viability and cervical mucus penetration. Contraception. 2003; 67(3):241-5. [DOI:10.1016/S0010-7824(02)00515-2]
Ypsilantis P, Papaioannou N, Psalla D, Politou M, Simopoulos C. Effects of single dose administration of ifosfamide on tests and semen characteristics in the rabbit. Reproductive Toxicology. 2003; 17(2):237-45. [DOI:10.1016/S0890-6238(02)00127-2]
Ypsilantis P, Papaioannou N, Psalla D, Politou M, Pitiakoudis M, Simopoulos C. Effects of subchronic ifosfamide-mesna treatment on testes and semen characteristics in the rabbit. Reproductive Toxicology. 2003; 17(6):699-708. [DOI:10.1016/j.reprotox.2003.08.003]
Xu DX, Shen HM, Zhu QX, Chua L, Wang QN, Chia SE, et al. The associations among semen quality, oxidative DNA damage in human spermatozoa and concentrations of cadmium, lead and selenium in seminal plasma. Mutation Research/Genetic Toxicology and Environmental Mutagenesis. 2003; 534(1-2):155-63. [DOI:10.1016/S1383-5718(02)00274-7]
Elbetieha A, Da’as SI, Khamas W, Darmani H. Evaluation of the toxic potentials of cypermethrin pesticide on some reproductive and fertility parameters in the male rats. Archives of Environmental Contamination and Toxicology. 2001; 41(4):522-8. [DOI:10.1007/s002440010280] [PMID]
Adelusi B, al-Twaijiri MH, al-Meshari A, Kangave D, al-Nuaim LA, Younnus B. Correlation of smoking and coffee drinking with sperm progressive motility in infertile males. The African journal of medical sciences. 1988; 27(1-2):47-50. [PMID]
Mita P, Piatti E, Romano A, Magro B. [Epidemiology of male infertility (Italian)]. Archivio Italiano di Urologia e Andrologia. 1998; 70(2):85-91. [PMID]
Wang WY, Thomas CMG, Merkus HMWM, Zielhuis GA, Doesburg WH, Steegers-Theunissen RPM. Cigarette smoking and the risk of male factor subfertility: minor association between cotinine in seminal plasma and semen morphology. Fertility and Sterility. 2000; 74(5):930-5. [DOI:10.1016/S0015-0282(00)01567-3]
Marinelli D, Gaspari L, Pedotti P, Taioli E. Mini-review of studies on the effect of smoking and drinking habits on semen parameters. International Journal of Hygiene and Environmental Health. 2004; 207(3):185-92. [DOI:10.1078/1438-4639-00283] [PMID]
Weathersbee PS, Lodge JR. Caffeine: its direct and indirect influence on reproduction. The Journal of Reproductive Medicine. 1977; 19(2):55-63. [PMID]
Klonoff-Cohen H, Bleha J, Lam-Kruglick P. A prospective study of the effects of female and male caffeine consumption on the reproductive endpoints of IVF and gamete intra-fallopian transfer. Human Reproduction. 2002; 17(7):1746-54. [DOI:10.1093/humrep/17.7.1746] [PMID]
Aritajat S, Kaweewat K, Manosroi J, Manosroi A. Dominant lethal test in rats treated with some plant extracts. The Southeast Asian Journal of Tropical Medicine and Public Health. 2000;31(Suppl. 1):171-3. [PMID]
Heidari M, Vaezi GH, Ameri F, Ghaffari-Novin M, Keramati K, Rajaei F. [Antiandrogenic effects of Rosmarinus officinalis extract on the reproductive tract of male rats (Persian)]. Tehran Univerasity Medical Journal. 2008; 65(Suppl. 3):26-32.
Jasemi M, Saki Gh, Rahim F. [The effect of Centella asiataica alcoholic extract on the serum levels of testesterone, FSH and LH in male wistar rat (Persian)]. Journal of Sabzevar university of medical sciences. 2009; 16(1):6-11.
Salamatmanesh M, Shiravi A, Heydari nasrabady M. [The effect of (Anethum graveolens) seed alcoholic extract on spermatogenesis in male wistar ratsccc. Journal of animal biolaogy. 2009; 1(2):23-30.
Kerishchi P, Kazem P, Rouhani A, Roostaeean A. [Effect of Achillea millefolium L. extract on spermatogenesis and HaG axis in adult BALB/C mice (Persian)]. Yafte. 2004; 6(22):13a8.
Ahmadi A, Nasiri Nejad F, Parivar K. [Effect of aqueaous axtract of the aerial part of the ruta graveolens on the spermatogenesis of immature Balb/C mice (Persian)]. Razi Journal of Medical Sciences. 2007; 14(56):13-20.
Mokhtari M, Shariati M, Gharamani R. [Effect of Trigonella foenum-graecum L. seed extract on concentration of tesatesterone and spermatogenesis in rats (Persian)]. Journal of Medicinal Plants. 2008; 7(1):12-20.
Modaresi M, Messripoor M, Asadi Morghmaleki M, Hamedanian MK. [The effect of Saffron extract on testis tissue (Persian)]. Iranian Journal of Medicinal and Aromatic Plants. 2008; 24(2):237-43.
Nouri M, Khaki A, Fathi Azar F, Rashidi MR. The protective effects of carrot seed extract on spermatogenesis and cauda epididymal sperm reserves in gentamicin treated rats. Yakhteh Medical Journal. 2009; 11(3):327-32.
Naseri M, Heydari nasrabadi M, Khodarahmi P, Ahmadi F, Mojibi P, Abotalebei H. [Study of the effect of Fumaria parviflora alcoholic extract on spermatogenesis in male rats (Persian)]. New Cellular and Molecular Biotechnology Journal. 2011; 1(2):61-5.
Mirfard M, Johari H, Mokhtari M, Hematkhah V, Jamali H, Allahverdi Gh. [The effect of hydro-alcoholic garlic extract on testis weight and spermatogenesis in mature male rats under chemotherapy with cyclophosphamide (Persian)]. Journal of Fasa University of Medical Sciences. 2011; 1(3):123-30.
Kazemi P, Jowhary H, Sharifi E, Zeraatpishe A. [Androgenic effect of Origanum vulgarel L. spp viride extract on hormone level of pituitary-gonadal axis in mature male vistar rats (Persian)]. Journal of Arak University of Medical Sciences. 2012; 14(6):89-96.
Hemayatkhah Jahromi V, Parivar K, Forozanfar M. The effect of cinnamon extract on spermatogenesis hormonal axis of pituitary gonad in mice. Iranian Journal of Applied Animal Science. 2011; 1(2):99-103.
Chauhan NS, Saraf DK, Dixita VK. Effect of vajikaran rasayana herbs on pituitary-gonadal axis. European Journal of Integrative Medicine. 2010; 2(2):89-91. [DOI:10.1016/j.eujim.2010.03.002]
Carlson BM. Human embryology and developmental biology. 3rd ed. Philadelphia: Mosby; 2004.
Cai L, Hales BF, Robaire B. Induction of apoptosis in the germ cells of adult male rats after exposure to cyclophosphamide. Biology of Reproduction. 1997; 56(6):1490-7. [DOI:10.1095/biolreprod56.6.1490] [PMID]
Nusier MK, Bataineh HN, Daradkah HM. Adverse effects of rosemary (Rosmarinus officinalis L.) on reproductive function in adult male rats. Experimental Biology and Medicine (Maywood). 2007; 232(6):809-13. [PMID]
Type of Study: Original |
Subject:
Reproductive Biology
Received: 2019/02/25 | Accepted: 2019/08/8 | Published: 2020/01/1
Received: 2019/02/25 | Accepted: 2019/08/8 | Published: 2020/01/1
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |